PR Newswire05.12.17
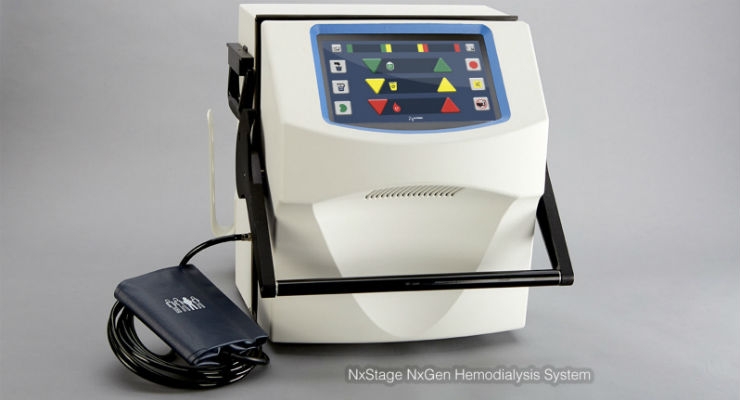
NxStage Medical Inc., a medical technology company focused on advancing renal care, announced that its next generation hemodialysis system has received FDA clearance. This innovative system consists of new features and capabilities, including a new touchscreen user interface and integrated blood pressure monitor designed to enhance ease of use for patients performing home hemodialysis.
"Early customer and patient feedback in the UK on our next generation system has been very positive, validating many of the benefits we expected," said Joseph E. Turk, president of NxStage Medical Inc. "We are very excited to bring these new features and capabilities that are designed to significantly improve our patients' therapy experience to the U.S. market. The touch screen interface provides an excellent platform to continue bringing new features and functionality moving forward."
The NxStage System One is the first and only truly portable hemodialysis system cleared specifically by the FDA for home hemodialysis and home nocturnal hemodialysis. The simplicity and revolutionary size (just over a foot tall) are intended to allow convenient use in patients' homes and give patients the freedom to travel with their therapy. When combined with the NxStage PureFlow SL Dialysis Preparation System, patients are able to further simplify, using ordinary tap water to create dialysis fluid on-site on-demand. Unlike conventional hemodialysis systems, the System One family of products requires no special infrastructure to operate. Under the guidance of their physician, patients can use the NxStage System One, with their trained partners, where, how and when it best meets their needs, including while they are sleeping—at home or on vacation and at a medically appropriate treatment frequency. In addition, NxStage's Nx2me Connected Health platform collects important NxStage System One and patient information for flexible viewing, monitoring and reporting that may improve patient management and patient retention, as well as simplify alternative site care. The System One is also used to provide a range of flexible therapy options in more traditional care settings such as hospitals and dialysis centers. Its safety and performance have been demonstrated by experience with more than 14 million treatments with over 30 thousand patients around the world.
"Early customer and patient feedback in the UK on our next generation system has been very positive, validating many of the benefits we expected," said Joseph E. Turk, president of NxStage Medical Inc. "We are very excited to bring these new features and capabilities that are designed to significantly improve our patients' therapy experience to the U.S. market. The touch screen interface provides an excellent platform to continue bringing new features and functionality moving forward."
The NxStage System One is the first and only truly portable hemodialysis system cleared specifically by the FDA for home hemodialysis and home nocturnal hemodialysis. The simplicity and revolutionary size (just over a foot tall) are intended to allow convenient use in patients' homes and give patients the freedom to travel with their therapy. When combined with the NxStage PureFlow SL Dialysis Preparation System, patients are able to further simplify, using ordinary tap water to create dialysis fluid on-site on-demand. Unlike conventional hemodialysis systems, the System One family of products requires no special infrastructure to operate. Under the guidance of their physician, patients can use the NxStage System One, with their trained partners, where, how and when it best meets their needs, including while they are sleeping—at home or on vacation and at a medically appropriate treatment frequency. In addition, NxStage's Nx2me Connected Health platform collects important NxStage System One and patient information for flexible viewing, monitoring and reporting that may improve patient management and patient retention, as well as simplify alternative site care. The System One is also used to provide a range of flexible therapy options in more traditional care settings such as hospitals and dialysis centers. Its safety and performance have been demonstrated by experience with more than 14 million treatments with over 30 thousand patients around the world.