Tactile Systems Technology Inc. 09.29.16
Tactile Systems Technology Inc., a developer and provider of medical devices for the treatment of chronic diseases at home, has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for the Flexitouch System in treating lymphedema of the head and neck, a common consequence of head and neck cancer.
“This is a major milestone for our company and a first for the lymphedema treatment market,” said Gerald R. Mattys, CEO of Tactile Medical. “More importantly, this new FDA clearance for our Flexitouch System represents a significant advancement in the treatment of head and neck lymphedema, as it enables patients to manage their condition at home. We look forward to the commercialization of our Flexitouch System for the treatment of head and neck lymphedema in the first half of 2017.”
Lymphedema is a chronic disease in which excess fluid accumulates in areas of the body when lymphatic vessels do not work efficiently or have been damaged. This chronic disease affects between 3 million and 5 million people in the United States. While there is no cure, lymphedema can be effectively managed to relieve symptoms, prevent worsening, and lower the risk of skin damage and infection.
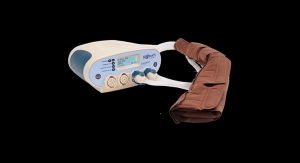
The Flexitouch System has been clinically proven to stimulate the lymphatic system. It delivers a noninvasive therapy that patients can administer themselves at home. Tens of thousands of patients with lymphedema in the limbs and trunk areas have benefited from the Flexitouch System since its introduction in 2002. The new FDA clearance expands the addressable patient population for the Flexitouch System to those that are suffering from lymphedema of the head and neck. The Flexitouch System is the first and only pneumatic compression device cleared for the treatment of lymphedema of the head and neck in the United States.
“It is widely known that lymphedema of the arms, legs or trunk causes immense suffering; utilizes health resources; and, as a result, is costly to the patient, community and society,” explained Alan T. Hirsch, M.D., Tactile Medical’s chief medical officer and director of the Vascular Medicine Program at the University of Minnesota Medical School. “Increasingly, we recognize head and neck lymphedema as being particularly challenging. This new at-home treatment of head and neck lymphedema will fill an important healthcare gap.”
Tactile Systems Technology Inc. develops and provides medical devices for the treatment of chronic diseases at home. The company is the sole manufacturer and distributor of the Flexitouch and Entré Systems, medical devices to help control symptoms of lymphedema, a chronic and progressive medical condition that is often an unintended consequence of cancer treatment, and the ACTitouch System, a medical device to treat venous leg ulcers and chronic venous insufficiency. The Minneapolis, Minn.-based company provides products for use both in the home and in health care institutions, including hospitals and vascular, wound and lymphedema clinics throughout the United States.
“This is a major milestone for our company and a first for the lymphedema treatment market,” said Gerald R. Mattys, CEO of Tactile Medical. “More importantly, this new FDA clearance for our Flexitouch System represents a significant advancement in the treatment of head and neck lymphedema, as it enables patients to manage their condition at home. We look forward to the commercialization of our Flexitouch System for the treatment of head and neck lymphedema in the first half of 2017.”
Lymphedema is a chronic disease in which excess fluid accumulates in areas of the body when lymphatic vessels do not work efficiently or have been damaged. This chronic disease affects between 3 million and 5 million people in the United States. While there is no cure, lymphedema can be effectively managed to relieve symptoms, prevent worsening, and lower the risk of skin damage and infection.
The Flexitouch System has been clinically proven to stimulate the lymphatic system. It delivers a noninvasive therapy that patients can administer themselves at home. Tens of thousands of patients with lymphedema in the limbs and trunk areas have benefited from the Flexitouch System since its introduction in 2002. The new FDA clearance expands the addressable patient population for the Flexitouch System to those that are suffering from lymphedema of the head and neck. The Flexitouch System is the first and only pneumatic compression device cleared for the treatment of lymphedema of the head and neck in the United States.
“It is widely known that lymphedema of the arms, legs or trunk causes immense suffering; utilizes health resources; and, as a result, is costly to the patient, community and society,” explained Alan T. Hirsch, M.D., Tactile Medical’s chief medical officer and director of the Vascular Medicine Program at the University of Minnesota Medical School. “Increasingly, we recognize head and neck lymphedema as being particularly challenging. This new at-home treatment of head and neck lymphedema will fill an important healthcare gap.”
Tactile Systems Technology Inc. develops and provides medical devices for the treatment of chronic diseases at home. The company is the sole manufacturer and distributor of the Flexitouch and Entré Systems, medical devices to help control symptoms of lymphedema, a chronic and progressive medical condition that is often an unintended consequence of cancer treatment, and the ACTitouch System, a medical device to treat venous leg ulcers and chronic venous insufficiency. The Minneapolis, Minn.-based company provides products for use both in the home and in health care institutions, including hospitals and vascular, wound and lymphedema clinics throughout the United States.