In 2015, Viveve announced that it entered into an exclusive distribution partnership for the Viveve System with NeoAsia PTE Ltd., a distributor of medical devices based in Singapore. The announcement of market approval in Singapore follows the recent announcement of regulatory approval of the Viveve System in South Korea.
"Asia represents a very important market and, we believe, a major commercial opportunity for Viveve. We have taken many steps to support rapid commercialization of the Viveve System in Asia, including establishing a relationship with our distribution partner, NeoAsia, in Singapore. The regulatory approvals for the Viveve System in Singapore and in South Korea are significant milestones in our efforts to make this clinically-proven treatment available to the millions of women throughout Asia and around the world who are living with vaginal laxity," said Patricia Scheller, CEO of Viveve.
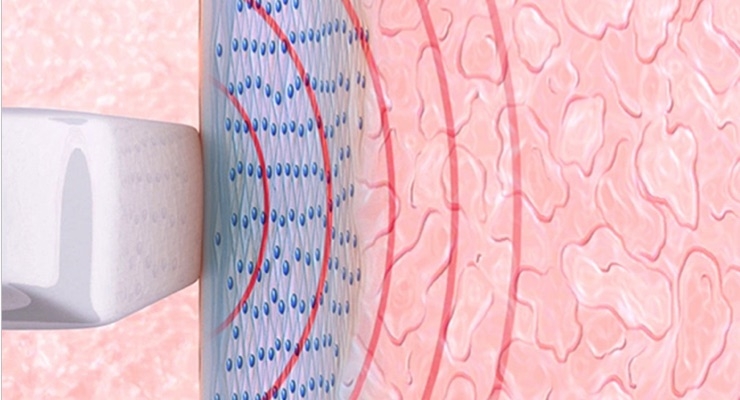
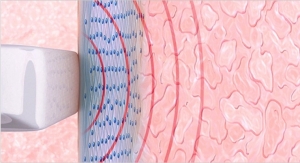
Viveve provides an office-based non-surgical, painless medical treatment for treating vaginal laxity. A physician performs the procedure using the Viveve System that works by cooling and gently heating vaginal tissue. This treatment option can increase feelings of vaginal tightness and improve sexual satisfaction.
Watch the video below for an animated demonstration of the Viveve treatment:
The Viveve Treatment is not cosmetic; instead, it works at the cellular level to rebuild and restore collagen and as a result can increase feelings of vaginal tightness and improve sexual satisfaction. The treatment can be completed in 30 minutes, is performed without anesthesia, and results in little to no downtime.
In two Viveve clinical studies, women who reported an increase in vaginal laxity following childbirth underwent the Viveve Treatment with the following results:
- Improved and sustained vaginal tightness was shown by 88 percent of the women at 12 months after treatment.
- Women who were treated experienced a 68 percent mean increase in their vaginal laxity scores at 12 months.
- Improvement in sexual satisfaction was maintained through 12 months after treatment.
- No serious adverse events were reported in either study.
Viveve Medical Inc. is a women's health company committed to advancing new solutions to improve women's overall well-being and quality of life. The Sunnyvale, Calif.-based company's lead product, the internationally patented Viveve System, is a non-surgical, non-ablative medical device that remodels collagen and restores tissue with only one treatment session. The Viveve System treats the condition of vaginal laxity that can result in decreased physical sensation and sexual satisfaction. Physician surveys indicate that vaginal laxity is the number one post-delivery physical change for women, being more prevalent than weight gain, urinary incontinence or stretch marks. The Viveve System has received regulatory approval in many countries throughout the world and is available through physician import license in Japan. It is currently not available for sale in the United States.