"We are very excited to commence this strategic relationship with GE Healthcare as we continue our efforts to optimize the supply chain for the Check-Cap system," said Bill Densel, CEO of Check-Cap. "GE Healthcare is a leader in the development, manufacturing, and distribution of diagnostic imaging agents and radio-pharmaceutical drugs and devices. We believe that leveraging their experience and expertise provides us with a significant opportunity to meet our goal of increasing the time and cost efficiencies of production of our capsule for use in future clinical trials and commercialization."
Colorectal cancer is the second leading cause of cancer death in the United States, with an estimated 134,000 diagnoses and 49,000 deaths in 2016. Despite compelling evidence that screening can detect colorectal cancer and precancerous polyps, nearly one-third of the recommended adult population has never been screened. The Check-Cap system was designed to improve the patient experience by eliminating the features of existing screening methods, such as bowel preparation, invasive procedure, and stool handling, that pose a barrier to test completion.
Check-Cap is currently conducting a multi-center clinical feasibility study and expects to file a CE Mark submission for the Check-Cap system in the first half of 2017.
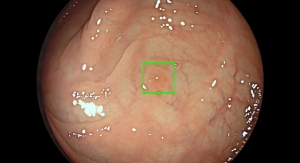
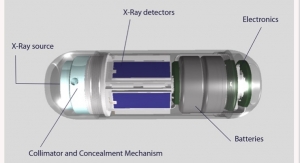
Check-Cap's ingestible capsule system uses ultra-low dose X-ray and wireless communication technologies to scan the inside of the colon as it moves naturally, while the patient follows his or her normal daily routine. After passage, the system generates a 3D map of the inner surface of the colon which enables detection of polyps and cancer. Designed to increase the willingness of individuals to participate in recommended colorectal cancer screening, the Check-Cap system addresses many frequently-cited barriers, including laxative bowel preparation, invasiveness, and sedation. The Check-Cap system is currently not cleared for marketing in any jurisdiction.
The idea of a prep-free, non-invasive manner to check for abnormalities in the colon has been gaining traction, especially as the first line method to screen for colorectal cancer. The hope is that non-invasive tests would boost the number of people who opt to get screened. Exact Sciences won U.S. Food and Drug Administration (FDA) approval for the first non-invasive DNA stool test for colorectal cancer called Cologuard in August 2014.
However, a more direct competitor to Check-Cap is Given Imaging’s PillCam, which is a similar swallowable imaging capsule that FDA approved in February 2014. Given Imaging was bought by Covidien in 2013 and is now part of Medtronic.
Last week, Israel's Office of the Chief Scientist awarded the Israeli firm a $1.25 million grant to help develop its technology and bring it to market. The company was founded in 2005.