Business Wire07.12.16
Intersect ENT Inc. has disclosed results of the Propel mini cohort of the PROGRESS study, a prospective, randomized, blinded, multi-center trial to assess the safety and efficacy of the PROPEL mini steroid releasing sinus stent when used following frontal sinus surgery with standard postoperative care.
The study met its primary efficacy endpoint, demonstrating a statistically significant 38 percent relative reduction in the need for post-operative interventions (p=0.007) at 30 days compared to surgery alone with standard post-operative care. Post-operative interventions included the need for additional surgical procedures and/or need for oral steroids. Clinical investigators also reported a 54 percemt relative reduction in restenosis, a 56 percent relative reduction in the need for oral steroids, and a 75 percent relative reduction in the need for surgical interventions on sides treated with Propel mini compared to surgery alone at 30 days. The device placement success rate was 100 percent and there were no device-related adverse events.
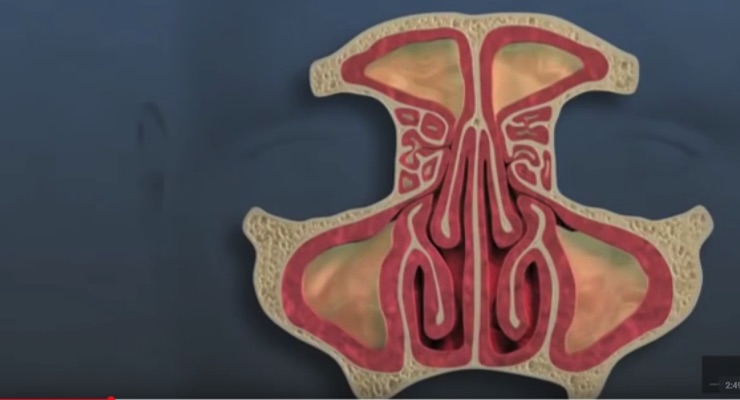
Watch the video below to discover how the Propel device works:
In March, Intersect ENT received approval for an expanded indication from the U.S. Food and Drug Administration (FDA) for placement of Propel mini in the frontal sinuses, which are located behind the eyebrows.
“The results from the PROGRESS study are important because they demonstrate that Propel mini improves the outcomes of frontal sinus surgery when added to standard postoperative care in chronic sinusitis patients with frontal sinus disease undergoing sinus surgery,” said Randall Ow, M.D., of Sacramento Ear, Nose, and Throat, a lead enroller in the study. “The recent expanded indication for Propel mini was much anticipated because the frontal sinus is more difficult to operate on and to manage post-operatively, compared to the other sinuses.”
“The positive outcomes from PROGRESS add to the clinical evidence demonstrating the benefits of steroid releasing implants,” said Lisa Earnhardt, president and CEO of Intersect ENT. “Frontal sinusitis affects as many as one in four patients undergoing surgery for chronic sinusitis. We are excited that this patient population now has access to treatment with Propel mini.”
Intersect ENT’s Propel and Propel mini are the first and only steroid releasing sinus implants approved by the FDA to maintain the open passages created in surgery, according to the company. The bioabsorbable products release mometasone furoate, an advanced steroid with anti-inflammatory properties, over time directly into the sinus lining, then fully dissolve. Propel’s effectiveness is supported by the highest level of clinical evidence, Level 1a, which demonstrates that Propel reduces inflammation and scarring after surgery, thereby lessening the need for post-operative surgical interventions and use of oral steroids. Both Propel and Propel mini are indicated for use following ethmoid sinus surgery. Additionally, Propel mini is indicated for use following frontal sinus surgery.
More than 100,000 U.S. patients have been treated with Propel and Propel mini, Intersect ENT executives claim.
Based in Menlo Park, Calif., Intersect ENT markets two steroid releasing implants, Propel and Propel mini, which have been clinically proven to improve surgical outcomes for chronic sinusitis patients undergoing sinus surgery. In addition, Intersect ENT is developing a pipeline of steroid releasing implants designed to provide ENT physicians with options to treat patients across the continuum of care for chronic sinusitis less invasively and more cost effectively. Chronic sinusitis is an inflammatory condition that can lead to debilitating symptoms and chronic infections, and is one of the most costly conditions to U.S. employers.
The study met its primary efficacy endpoint, demonstrating a statistically significant 38 percent relative reduction in the need for post-operative interventions (p=0.007) at 30 days compared to surgery alone with standard post-operative care. Post-operative interventions included the need for additional surgical procedures and/or need for oral steroids. Clinical investigators also reported a 54 percemt relative reduction in restenosis, a 56 percent relative reduction in the need for oral steroids, and a 75 percent relative reduction in the need for surgical interventions on sides treated with Propel mini compared to surgery alone at 30 days. The device placement success rate was 100 percent and there were no device-related adverse events.
Watch the video below to discover how the Propel device works:
In March, Intersect ENT received approval for an expanded indication from the U.S. Food and Drug Administration (FDA) for placement of Propel mini in the frontal sinuses, which are located behind the eyebrows.
“The results from the PROGRESS study are important because they demonstrate that Propel mini improves the outcomes of frontal sinus surgery when added to standard postoperative care in chronic sinusitis patients with frontal sinus disease undergoing sinus surgery,” said Randall Ow, M.D., of Sacramento Ear, Nose, and Throat, a lead enroller in the study. “The recent expanded indication for Propel mini was much anticipated because the frontal sinus is more difficult to operate on and to manage post-operatively, compared to the other sinuses.”
“The positive outcomes from PROGRESS add to the clinical evidence demonstrating the benefits of steroid releasing implants,” said Lisa Earnhardt, president and CEO of Intersect ENT. “Frontal sinusitis affects as many as one in four patients undergoing surgery for chronic sinusitis. We are excited that this patient population now has access to treatment with Propel mini.”
Intersect ENT’s Propel and Propel mini are the first and only steroid releasing sinus implants approved by the FDA to maintain the open passages created in surgery, according to the company. The bioabsorbable products release mometasone furoate, an advanced steroid with anti-inflammatory properties, over time directly into the sinus lining, then fully dissolve. Propel’s effectiveness is supported by the highest level of clinical evidence, Level 1a, which demonstrates that Propel reduces inflammation and scarring after surgery, thereby lessening the need for post-operative surgical interventions and use of oral steroids. Both Propel and Propel mini are indicated for use following ethmoid sinus surgery. Additionally, Propel mini is indicated for use following frontal sinus surgery.
More than 100,000 U.S. patients have been treated with Propel and Propel mini, Intersect ENT executives claim.
Based in Menlo Park, Calif., Intersect ENT markets two steroid releasing implants, Propel and Propel mini, which have been clinically proven to improve surgical outcomes for chronic sinusitis patients undergoing sinus surgery. In addition, Intersect ENT is developing a pipeline of steroid releasing implants designed to provide ENT physicians with options to treat patients across the continuum of care for chronic sinusitis less invasively and more cost effectively. Chronic sinusitis is an inflammatory condition that can lead to debilitating symptoms and chronic infections, and is one of the most costly conditions to U.S. employers.