Polyganics06.21.16
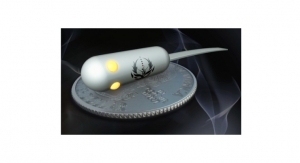
Polyganics, a privately held medical technology company focused on the commercialization of proprietary products and technologies for peripheral nerve reconstruction and regeneration, has announced that it has received the CE Mark for Neurocap, its nerve capping device. Polyganics intends to launch Neurocap in several European countries later this year.
Neurocap is an absorbable implant for the treatment and reduction of symptomatic neuroma in peripheral nerves. The device was cleared for sales in the United States in January 2016, and Polyganics introduced Neurocap during the Annual Meeting of the American Society of Surgery of the Hand in Phoenix last January.
In February, the European STOP NEUROMA study started to gather evidence for the long-term effectiveness of Neurocap in the reduction of painful neuroma formation. Coordinating Investigator Mariëtta Bertleff, MD, has successfully enrolled the first patients at the MC Groep hospital in Emmeloord, The Netherlands. Through this study, Polyganics is collecting more data on the clinical performance of Neurocap’s ability to isolate the nerve end, and the product’s effectiveness with respect to the reduction of pain from the symptomatic neuroma and prevention of pain reoccurrence.
Rudy Mareel, CEO of Polyganics said, “CE regulatory approval is a key milestone for our nerve capping device. We strongly believe Neurocap represents an important addition to the surgeons’ tool-box in the treatment of peripheral nerve injuries enhancing surgical outcomes and patient recovery.”
“Furthermore, if Neurocap shows effectiveness in terms of preventing pain symptoms to return over a one-year period, the device could be used in the prevention of painful neuroma formation following amputations. This is an additional, even more significant population which is steadily increasing due to the growing prevalence of diseases such as diabetes,” he added.
A delegation from Polyganics, including Rudy Mareel, CEO, Romke Ribbels, Director Marketing & Business Development and Raoul Beex, Director Sales, will be attending the Federation of European Societies for Surgery of the Hand (FESSH) on 22-25 June in Santander, Spain and will be present at Exhibitor Booth 21 throughout the conference.
Neurocap is an absorbable implant for the treatment and reduction of symptomatic neuroma in peripheral nerves. The device was cleared for sales in the United States in January 2016, and Polyganics introduced Neurocap during the Annual Meeting of the American Society of Surgery of the Hand in Phoenix last January.
In February, the European STOP NEUROMA study started to gather evidence for the long-term effectiveness of Neurocap in the reduction of painful neuroma formation. Coordinating Investigator Mariëtta Bertleff, MD, has successfully enrolled the first patients at the MC Groep hospital in Emmeloord, The Netherlands. Through this study, Polyganics is collecting more data on the clinical performance of Neurocap’s ability to isolate the nerve end, and the product’s effectiveness with respect to the reduction of pain from the symptomatic neuroma and prevention of pain reoccurrence.
Rudy Mareel, CEO of Polyganics said, “CE regulatory approval is a key milestone for our nerve capping device. We strongly believe Neurocap represents an important addition to the surgeons’ tool-box in the treatment of peripheral nerve injuries enhancing surgical outcomes and patient recovery.”
“Furthermore, if Neurocap shows effectiveness in terms of preventing pain symptoms to return over a one-year period, the device could be used in the prevention of painful neuroma formation following amputations. This is an additional, even more significant population which is steadily increasing due to the growing prevalence of diseases such as diabetes,” he added.
A delegation from Polyganics, including Rudy Mareel, CEO, Romke Ribbels, Director Marketing & Business Development and Raoul Beex, Director Sales, will be attending the Federation of European Societies for Surgery of the Hand (FESSH) on 22-25 June in Santander, Spain and will be present at Exhibitor Booth 21 throughout the conference.