Business Wire05.31.16
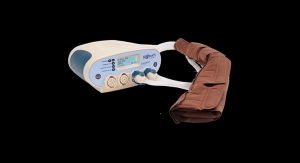
EndoGastric Solutions (EGS) announced the first use of the recently cleared EsophyX Z Fastener Delivery System (EsophyX Z device) during a transoral incisionless fundoplication (TIF) procedure at the University of California-Irvine Health.
Interventional gastroenterologist Kenneth Chang, M.D., executive director of the UC Irvine Health H.H. Chao Comprehensive Digestive Disease Center, performed the TIF procedure using the new EsophyX Z device, which received U.S. Food and Drug Administration (FDA) 510(k) clearance earlier this month.
“There are certain GERD patients who don’t respond effectively to medical management with proton pump inhibitors,” said Chang. “Long-term data continue to demonstrate the positive impact the TIF procedure for reflux has in controlling chronic, troublesome GERD symptoms.”
The TIF procedure using the EsophyX Z device allows physicians to reconstruct the gastroesophageal valve by performing a partial fundoplication, transorally, and securing the gastric tissue with fewer procedural steps.
“We are excited and honored to have Dr. Chang perform the first TIF procedure using the new EsophyX Z device via the TIF 2.0 procedure to treat a chronic GERD patient,” said Skip Baldino, president and CEO of EndoGastric Solutions. “This marks another milestone for EGS as we continue to address the significant problem of GERD and focus on filling the treatment gap between medication and more invasive surgery.”
EsophyX Z, designed for compatibility with the company’s existing 7.5 mm SerosaFuse Implantable Fastener cartridges, will enter a limited U.S. release before full commercialization during the summer of 2016. The device will also be used in the American Gastroenterological Association’s STAR Registry, which will provide the first real-world data comparing patient outcomes after traditional laparoscopic surgery and TIF procedure with the EsophyX device.
The original EsophyX device was cleared by the FDA in 2007. EGS launched the third generation EsophyX device, the EsophyX Z, in 2015. The technology has continued to evolve and is a clinically backed tool for physician use in the treatment of gastroesophageal reflux disease (GERD). The EsophyX technology now enables surgeons and gastroenterologists to use a wider selection of endoscopes—including low profile and larger high-definition models—to treat the underlying anatomical cause of GERD. The EsophyX technology is used to reconstruct the gastroesophageal valve (GEV) and restore its function as a barrier, preventing stomach acids refluxing back into the esophagus. The device is inserted through the patient’s mouth with direct visual guidance from an endoscope.
The EsophyX device with SerosaFuse fasteners is indicated for use in transoral tissue approximation, full thickness plication and ligation in the gastrointestinal tract and is indicated for the treatment of symptomatic chronic gastroesophageal reflux disease in patients who require and respond to pharmacological therapy. It is also indicated to narrow the gastroesophageal junction and reduce hiatal hernia ≤ 2cm in size in patients with symptomatic chronic gastroesophageal reflux disease.
Performed without the need for external incisions through the skin, the TIF procedure offers patients who require an anatomical repair another treatment option to correct the underlying cause of GERD. Studies show that for up to three years after the TIF procedure esophageal inflammation (esophagitis) is eliminated and most patients are able to stop using daily PPI medications to control symptoms.
More than 17,000 TIF patients have been treated worldwide since EsophyX device clearance in 2007. More than 60 peer-review papers from more than 50 centers have been published documenting consistent outcomes on over 1,100 unique study patients.
Gastroesophageal reflux disease is a chronic condition in which the gastroesophageal valve (GEV) allows gastric contents to reflux (wash backwards) into the esophagus, causing heartburn and possible injury to the esophageal lining. The stomach produces hydrochloric acid and other digestive enzymes after a meal to aid in the digestion of food. The cells that line the stomach are coated with a protective mucus that can withstand gastric contents, while the cells that line the esophagus lack the same protection.
GERD is the most common gastrointestinal-related diagnosis made by physicians during clinical visits in the United States. It is estimated that pain and discomfort from acid reflux impacts more than 80 million people at least once per month in the United States. The standard recommendations for symptomatic GERD patients include lifestyle changes (e.g., diet, scheduled eating times, and sleeping positions) and escalating doses of prescription medications for prolonged periods of time. Long-term, maximum-dose usage of prescription medications has been linked to a variety of other health complications.
Based in Redmond, Wash., EndoGastric Solutions develops and commercializes evidence-based, incisionless surgical technology for the treatment of gastroesophageal reflux disease.
Interventional gastroenterologist Kenneth Chang, M.D., executive director of the UC Irvine Health H.H. Chao Comprehensive Digestive Disease Center, performed the TIF procedure using the new EsophyX Z device, which received U.S. Food and Drug Administration (FDA) 510(k) clearance earlier this month.
“There are certain GERD patients who don’t respond effectively to medical management with proton pump inhibitors,” said Chang. “Long-term data continue to demonstrate the positive impact the TIF procedure for reflux has in controlling chronic, troublesome GERD symptoms.”
The TIF procedure using the EsophyX Z device allows physicians to reconstruct the gastroesophageal valve by performing a partial fundoplication, transorally, and securing the gastric tissue with fewer procedural steps.
“We are excited and honored to have Dr. Chang perform the first TIF procedure using the new EsophyX Z device via the TIF 2.0 procedure to treat a chronic GERD patient,” said Skip Baldino, president and CEO of EndoGastric Solutions. “This marks another milestone for EGS as we continue to address the significant problem of GERD and focus on filling the treatment gap between medication and more invasive surgery.”
EsophyX Z, designed for compatibility with the company’s existing 7.5 mm SerosaFuse Implantable Fastener cartridges, will enter a limited U.S. release before full commercialization during the summer of 2016. The device will also be used in the American Gastroenterological Association’s STAR Registry, which will provide the first real-world data comparing patient outcomes after traditional laparoscopic surgery and TIF procedure with the EsophyX device.
The original EsophyX device was cleared by the FDA in 2007. EGS launched the third generation EsophyX device, the EsophyX Z, in 2015. The technology has continued to evolve and is a clinically backed tool for physician use in the treatment of gastroesophageal reflux disease (GERD). The EsophyX technology now enables surgeons and gastroenterologists to use a wider selection of endoscopes—including low profile and larger high-definition models—to treat the underlying anatomical cause of GERD. The EsophyX technology is used to reconstruct the gastroesophageal valve (GEV) and restore its function as a barrier, preventing stomach acids refluxing back into the esophagus. The device is inserted through the patient’s mouth with direct visual guidance from an endoscope.
The EsophyX device with SerosaFuse fasteners is indicated for use in transoral tissue approximation, full thickness plication and ligation in the gastrointestinal tract and is indicated for the treatment of symptomatic chronic gastroesophageal reflux disease in patients who require and respond to pharmacological therapy. It is also indicated to narrow the gastroesophageal junction and reduce hiatal hernia ≤ 2cm in size in patients with symptomatic chronic gastroesophageal reflux disease.
Performed without the need for external incisions through the skin, the TIF procedure offers patients who require an anatomical repair another treatment option to correct the underlying cause of GERD. Studies show that for up to three years after the TIF procedure esophageal inflammation (esophagitis) is eliminated and most patients are able to stop using daily PPI medications to control symptoms.
More than 17,000 TIF patients have been treated worldwide since EsophyX device clearance in 2007. More than 60 peer-review papers from more than 50 centers have been published documenting consistent outcomes on over 1,100 unique study patients.
Gastroesophageal reflux disease is a chronic condition in which the gastroesophageal valve (GEV) allows gastric contents to reflux (wash backwards) into the esophagus, causing heartburn and possible injury to the esophageal lining. The stomach produces hydrochloric acid and other digestive enzymes after a meal to aid in the digestion of food. The cells that line the stomach are coated with a protective mucus that can withstand gastric contents, while the cells that line the esophagus lack the same protection.
GERD is the most common gastrointestinal-related diagnosis made by physicians during clinical visits in the United States. It is estimated that pain and discomfort from acid reflux impacts more than 80 million people at least once per month in the United States. The standard recommendations for symptomatic GERD patients include lifestyle changes (e.g., diet, scheduled eating times, and sleeping positions) and escalating doses of prescription medications for prolonged periods of time. Long-term, maximum-dose usage of prescription medications has been linked to a variety of other health complications.
Based in Redmond, Wash., EndoGastric Solutions develops and commercializes evidence-based, incisionless surgical technology for the treatment of gastroesophageal reflux disease.