“We are excited to fill this critical role in the organization with such high-caliber talent as Alex. He has led the successful adoption of several transformational products throughout his career in the structural heart space and is exactly the leader NeoChord needs as we expand our commercial footprint with the DS1000 System in Europe,” said David Chung, president and CEO.
Most recently, Jonkers was with Medtronic plc, leading a large commercial organization responsible for catheter-based therapies and surgical heart valves across the Europe, Middle East and Africa region. Prior to Medtronic, he was with CoreValve Inc., serving in a clinical and sales leadership capacity and a key member of the team that directed the successful integration of CoreValve into the Medtronic organization upon acquisition.
“I am thrilled to be a part of the team at NeoChord and am excited to bring this innovative procedure option to more patients,” said Jonkers. “As an off-bypass device, I see tremendous potential for consideration of NeoChord’s technology as an early treatment option for patients suffering from degenerative mitral valve regurgitation (DMR).”
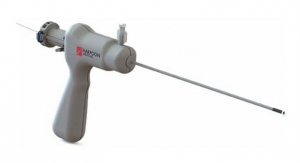
Additionally, the company today announced the treatment of more than 400 patients with the DS1000 System. The NeoChord DS1000 is a disposable device that is intended to replace damaged chordae by delivering artifical chordae tendinae or "neochords" in a beating heart using minimally invasive techniques. In patients with severe degenerative mitral valve regurgitation, early surgery is recommended for patients with no symptoms because the probability of surgical success is high. Waiting until symptoms develop is associated with harmful changes in the heart’s structure and function that may increase the potential for surgical complications. However, patients with no symptoms might be reluctant to undergo open-heart surgery with bypass. The NeoChord DS1000 may offer patients a less invasive procedure choice.
The NeoChord procedure is performed on the beating heart through a 2 to 3 inch incision between the ribs, unlike some repair procedures where the sternum is cut and the rib cage is spread open. Using echocardiographic guidance, the NeoChord DS1000 is introduced through the lowest part of the heart (apex), into the left ventricle, and between the mitral valve leaflets. The prolapsed leaflet is then grasped using the expandable jaws of the device. When the monitor confirms that the leaflet has been adequately captured, the ePTFE suture is deployed and attached to the leaflet. The suture is then pulled through the apex of the heart as the DS1000 is removed. The correct length of the suture is determined by using real time echocardiographic guidance and observing the improvement in mitral valve regurgitation in the beating heart. The suture is then secured to the apex of the heart.
“Achieving this milestone is a significant endorsement of the technology and therapy from the cardiovascular community,” said Chung.Based in St. Louis Park, Minn., NeoChord is a privately held developer of technology for the minimally invasive repair of degenerative mitral regurgitation (DMR), a progressive disease that can result in atrial fibrillation, congestive heart failure, and death when left untreated. NeoChord received CE market clearance in December 2012 for the DS1000 System to treat DMR without the use of cardiopulmonary bypass. The device is limited to investigational use in the United States.