11.10.15
NanoVibronix Inc. has released its hands-free ultrasound therapy device in the United States.
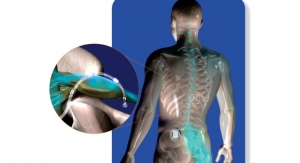
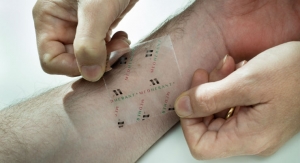
The U.S. Food and Drug Administration has granted the company 510(k) clearance for its PainShield MD, a patch-based device that uses ultrasound technology to treat pain and promote soft tissue healing. For decades, ultrasound therapy has been acknowledged as a valuable tool for pain relief and tissue healing. The PainShield MD consists of a disposable patch connected to a portable reusable driver that propagates therapeutic ultrasound waves to the desired area of the body.
"PainShield MD, which implements our unique low frequency, low-intensity ultrasound platform, is the first product from our pipeline to be cleared for marketing in the U.S. and represents a major milestone for NanoVibronix," said NanoVibronix President/CEO William Stern, Ph.D. "We have created a platform that changes the way pain, the most common medical complaint, is managed. Our portable device can treat pain locally including trigeminal neuralgia, chronic pelvic pain, tendonitis, musculoskeletal disorders, and post operative pain."
Based in Nesher, Israel, NanoVibronix develops products that incorporate its proprietary low-intensity acoustic technology. These therapeutic ultrasound devices are being used in various medical applications for pain relief and wound healing, according to executives. The technology also is being used as an attachment for indwelling urinary catheters to decrease bacterial growth, biofilm formation and urethral trauma while improving patient comfort.
The U.S. Food and Drug Administration has granted the company 510(k) clearance for its PainShield MD, a patch-based device that uses ultrasound technology to treat pain and promote soft tissue healing. For decades, ultrasound therapy has been acknowledged as a valuable tool for pain relief and tissue healing. The PainShield MD consists of a disposable patch connected to a portable reusable driver that propagates therapeutic ultrasound waves to the desired area of the body.
"PainShield MD, which implements our unique low frequency, low-intensity ultrasound platform, is the first product from our pipeline to be cleared for marketing in the U.S. and represents a major milestone for NanoVibronix," said NanoVibronix President/CEO William Stern, Ph.D. "We have created a platform that changes the way pain, the most common medical complaint, is managed. Our portable device can treat pain locally including trigeminal neuralgia, chronic pelvic pain, tendonitis, musculoskeletal disorders, and post operative pain."
Based in Nesher, Israel, NanoVibronix develops products that incorporate its proprietary low-intensity acoustic technology. These therapeutic ultrasound devices are being used in various medical applications for pain relief and wound healing, according to executives. The technology also is being used as an attachment for indwelling urinary catheters to decrease bacterial growth, biofilm formation and urethral trauma while improving patient comfort.