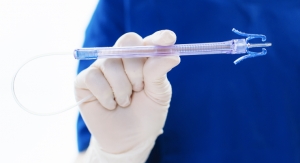
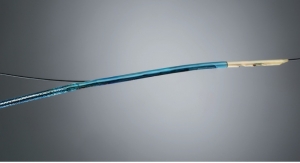
Velano Vascular has received CE Mark certification for its blood-draw technology.
The product is meant to be an alternative to conventional blood drawing in hospitals, a process that relies on needle sticks and can require the use of more than one for successful blood specimen collection.
Instead, the device uses a peripheral intravenous catheter, which most hospitalized patients already have in place, to gather blood, company executives said. The innovation is expected to improve patient experience and medical professional safety.
The devicemaker has begun clinical trials in the United tates,. where the product was U.S. Food and Drug Administration 510(k)-cleared in February. In addition, the company is collaborating with Brigham and Women’s Hospital in Boston, Mass.
"This important regulatory approval underscores the need for this technology around the world," said Pitou Devgon, M.D., Velano Vascular's co-founder and president. "Our aim is to improve the way medicine has been practiced for decades in order to improve the patient experience and increase safety for medical professionals. Receiving CE Mark moves us one significant step closer to that goal."
Founded by a patient advocate and a physician, Velano Vascular is backed by First Round Capital, Kapor Capital, Safeguard Scientifics, White Owl Capital, Griffin Hospital (Derby, Conn.), The Children's Hospital of Philadelphia, and several health-industry veterans. Velano Vascular's clinical collaborators include several hospital systems in the United States. The company is based in Philadelphia, Pa., and San Francisco, Calif.