11.02.15
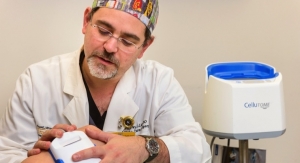
Columbia, Md.-based Osiris Therapeutics Inc., a cellular regenerative medicine company focused on products to treat conditions in wound care, orthopedics and sports medicine, has launched Truskin, a cryopreserved, living skin allograft.
Truskin has been developed to provide healthcare professionals a new option for treating large, chronic wounds in the hospital outpatient, private office, and/or surgical settings, where allograft options are limited and reimbursement pressures are high. Specifically in the hospital outpatient setting, it has become common to relegate product usage to those products that are under their assigned bundled payment amounts. Currently, patients with larger wounds are limited in the offerings of advanced skin substitutes. Osiris’ strategy is to provide a larger-sized, high quality graft that fits within the Medicare bundled payment amounts to ensure that providers have an Osiris product option despite wound size. Osiris’ large Truskin allograft are hoped to ensure that physicians have a larger-sized, high quality graft for patients regardless of wound size.
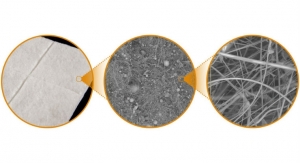
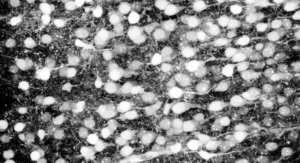
Characterization studies demonstrate that the new proprietary Truskin process maintains key structural and functional properties of fresh skin allografts to promote proper wound closure. Consistent with its evidence-based approach, Osiris plans to introduce a robust scientific and clinical program for Truskin, including multiple randomized controlled trials, comparative studies, case studies, and clinical and scientific posters and abstracts.
“Osiris is committed to developing products that address unmet medical needs,” said Alla Danilkovitch, Ph.D., chief scientific officer. “This new skin allograft, Truskin, provides structural and functional properties similar to fresh skin that are essential for the repair of chronic wounds in patients.”
Truskin has been developed to provide healthcare professionals a new option for treating large, chronic wounds in the hospital outpatient, private office, and/or surgical settings, where allograft options are limited and reimbursement pressures are high. Specifically in the hospital outpatient setting, it has become common to relegate product usage to those products that are under their assigned bundled payment amounts. Currently, patients with larger wounds are limited in the offerings of advanced skin substitutes. Osiris’ strategy is to provide a larger-sized, high quality graft that fits within the Medicare bundled payment amounts to ensure that providers have an Osiris product option despite wound size. Osiris’ large Truskin allograft are hoped to ensure that physicians have a larger-sized, high quality graft for patients regardless of wound size.
Characterization studies demonstrate that the new proprietary Truskin process maintains key structural and functional properties of fresh skin allografts to promote proper wound closure. Consistent with its evidence-based approach, Osiris plans to introduce a robust scientific and clinical program for Truskin, including multiple randomized controlled trials, comparative studies, case studies, and clinical and scientific posters and abstracts.
“Osiris is committed to developing products that address unmet medical needs,” said Alla Danilkovitch, Ph.D., chief scientific officer. “This new skin allograft, Truskin, provides structural and functional properties similar to fresh skin that are essential for the repair of chronic wounds in patients.”