There apparently is some validity to women's complaints about Bayer AG's Essure device.
A new study from Weill Cornell Medicine concludes that recipients of the birth control product have a 10-fold higher risk of undergoing additional surgeries in the first year after the initial procedure.
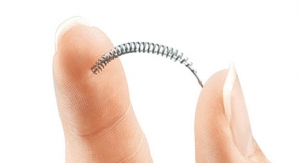
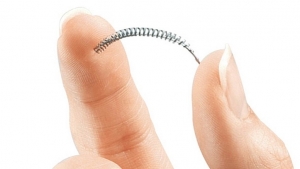
The study is the first to compare the efficacy of laparoscopic sterilization, a procedure that obstructs the fallopian tubes, with a nonsurgical method that uses the implant Essure contraceptive device, a metal spring made from a nickel-titanium alloy, to block the tubes. The Weill Cornell Medicine team found the risk of post-procedure operation for women who opt for hysteroscopic sterilization was 10 times higher than for women who underwent laparoscopic sterilization.
Using New York State patient information, the authors found that nearly one-fourth of the estimated 600,000 women nationwide who underwent a permanent birth control procedure chose hysteroscopic sterilization.
"We found tenfold higher risk of repeat surgery after Essure when compared to laparoscopic surgery. This means that nationally more than 10,000 women likely underwent additional surgery in the past five years," said Art Sedrakyan, M.D., Ph.D., a professor of healthcare policy and research at Weill Cornell Medicine, who led the study, published Oct. 13 in the BMJ.
In hysteroscopic sterilization, the metal Essure device is inserted into the patient's fallopian tubes. In time, scar tissue grows around the device, forming a barrier that prevents eggs from becoming fertilized. Bayer claims the procedure takes about 10 minutes, and doesn't require general anesthesia.
Patients, however, recently have filed lawsuits against Bayer, claiming the implant has led to allergic reactions to nickel, severe pelvic pain and post-procedure operations, including hysterectomies and attempts to stabilize the Essure device after it has inappropriately shifted.
The U.S. Food and Drug Administration is reviewing complaints about the safety of the device, but the Weill Cornell Medicine study represents the first analysis of 30-day, and one-, two- and three-year outcomes of 50,000 patients in New York undergoing permanent birth control procedures.
"In our study, we found an over 10-fold higher risk of reoperation associated with hysteroscopic sterilization with the Essure device compared to laparoscopic sterilization, translating into about 21 additional reoperations per 1,000 patients undergoing surgery. Meanwhile, the occurrence of unintended pregnancy was not different in the two groups," the study authors wrote.
The Weill Cornell Medicine researchers recommended deeper communication between physician and patient regarding the risks associated with the device, plus the creation of national and state registries to track specific safety issues related to the Essure device.
"Establishing registries can help us elucidate specific complications that lead to repeat surgeries and better understand if it is possible to overcome technological limitations that cause these device failures," Sedrakyan said. "We hope this publication and dissemination will lead to appropriate action by decision makers."