10.09.15
W. L. Gore & Associates Inc. (Gore) has received approval from the United States and Japan to begin clinical trials for its Excluder Conformable AAA Endoprosthesis.
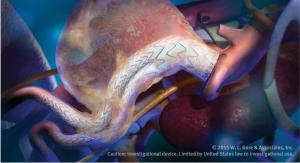
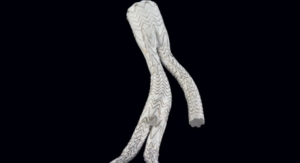
The blessing from the U.S. Food and Drug Administration and Japan's Pharmaceuticals and Medical Devices Agency allows the company to study the safety and effectiveness of the endoprosthesis device in treating infrarenal abdominal aortic aneurysms (AAA) with challenging patient anatomy. The new device is the next generation of the Gore Excluder AAA Endoprosthesis and includes several key design features, including increased conformability and a delivery system with angulation control.
Robert Rhee, M.D., Chief of Vascular and Endovascular Surgery at Maimonides Medical Center in New York, N.Y., will be the principal investigator for the United States, and Takao Ohki M.D., Ph.D., chairman and professor of the Department of Surgery at Jikei University School of Medicine in Tokyo, will be the principal investigator for Japan.
“Extreme proximal neck anatomies are one of the main reasons why a AAA patient would not qualify for endovascular therapy,” said Ohki. “The new Gore Excluder Conformable AAA Endoprosthesis will provide a minimally invasive treatment option for patients in this situation, some of whom are unfit for open surgery. For these patients, this device may be their only treatment option.”
The clinical trial consists of two sub-studies, each assessing the device for a different range of patient anatomies. The first sub-study will assess the device in proximal aortic neck angles of zero to 60 degrees and aortic neck lengths of 10 millimeters or greater. The other sub-study will evaluate proximal aortic neck angles of 61 to 90 degrees and aortic neck lengths of 10 millimeters or greater. If approved, the Gore Excluder Conformable AAA Endoprosthesis would be the only device indicated for aortic neck angles up to 90 degrees and aortic neck lengths as short as 10 millimeters.
The Gore Excluder Endoprosthesis is deployed through an enhanced delivery system that includes angulation control, giving physicians the option to angle or bend the device to achieve orthogonal placement to the aortic blood flow lumen and to maximize the conformability of the device, according to the company. Like the Gore C3 Delivery System, the Gore Excluder Endoprosthesis delivery system gives doctors the ability to reposition the device after initial deployment if needed to achieve optimal device placement.
“Even if a device is flexible enough to accommodate a highly angulated aortic neck, achieving conformability during deployment with current delivery systems can be difficult,” said Rhee. “The Excluder Conformable device delivery system is designed to give physicians a greater level of control over device placement to maximize device conformability regardless of anatomic challenges.”
The Excluder AAA Endoprosthesis builds on the clinical performance of the Gore Excluder AAA Device, supported by 17 years of worldwide experience and distribution of more than 215,000 devices.
“We’ve combined a novel stent design, device conformability, and an enhanced delivery system to give physicians even more control during the procedure,” said Ryan Takeuchi, aortic business unit leader at Gore. “We feel this combination is critically important to effectively expand the treatable population, affording more patients the benefits of endovascular therapy. We are eager to begin this clinical study in both the U.S. and Japan.”
W.L. Gore & Associates is headquartered in Flagstaff, Ariz.
The blessing from the U.S. Food and Drug Administration and Japan's Pharmaceuticals and Medical Devices Agency allows the company to study the safety and effectiveness of the endoprosthesis device in treating infrarenal abdominal aortic aneurysms (AAA) with challenging patient anatomy. The new device is the next generation of the Gore Excluder AAA Endoprosthesis and includes several key design features, including increased conformability and a delivery system with angulation control.
Robert Rhee, M.D., Chief of Vascular and Endovascular Surgery at Maimonides Medical Center in New York, N.Y., will be the principal investigator for the United States, and Takao Ohki M.D., Ph.D., chairman and professor of the Department of Surgery at Jikei University School of Medicine in Tokyo, will be the principal investigator for Japan.
“Extreme proximal neck anatomies are one of the main reasons why a AAA patient would not qualify for endovascular therapy,” said Ohki. “The new Gore Excluder Conformable AAA Endoprosthesis will provide a minimally invasive treatment option for patients in this situation, some of whom are unfit for open surgery. For these patients, this device may be their only treatment option.”
The clinical trial consists of two sub-studies, each assessing the device for a different range of patient anatomies. The first sub-study will assess the device in proximal aortic neck angles of zero to 60 degrees and aortic neck lengths of 10 millimeters or greater. The other sub-study will evaluate proximal aortic neck angles of 61 to 90 degrees and aortic neck lengths of 10 millimeters or greater. If approved, the Gore Excluder Conformable AAA Endoprosthesis would be the only device indicated for aortic neck angles up to 90 degrees and aortic neck lengths as short as 10 millimeters.
The Gore Excluder Endoprosthesis is deployed through an enhanced delivery system that includes angulation control, giving physicians the option to angle or bend the device to achieve orthogonal placement to the aortic blood flow lumen and to maximize the conformability of the device, according to the company. Like the Gore C3 Delivery System, the Gore Excluder Endoprosthesis delivery system gives doctors the ability to reposition the device after initial deployment if needed to achieve optimal device placement.
“Even if a device is flexible enough to accommodate a highly angulated aortic neck, achieving conformability during deployment with current delivery systems can be difficult,” said Rhee. “The Excluder Conformable device delivery system is designed to give physicians a greater level of control over device placement to maximize device conformability regardless of anatomic challenges.”
The Excluder AAA Endoprosthesis builds on the clinical performance of the Gore Excluder AAA Device, supported by 17 years of worldwide experience and distribution of more than 215,000 devices.
“We’ve combined a novel stent design, device conformability, and an enhanced delivery system to give physicians even more control during the procedure,” said Ryan Takeuchi, aortic business unit leader at Gore. “We feel this combination is critically important to effectively expand the treatable population, affording more patients the benefits of endovascular therapy. We are eager to begin this clinical study in both the U.S. and Japan.”
W.L. Gore & Associates is headquartered in Flagstaff, Ariz.