09.10.15
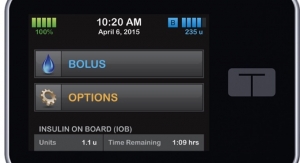
San Diego, Calif.-based Tandem Diabetes Care Inc. has earned U.S. Food and Drug Administration (FDA) approval for its t:slim G4 insulin pump. The device is a touch-screen pump with continuous glucose monitoring (CGM) integration, for use by people 12 years of age or older who use insulin. It combines features of the company’s t:slim insulin pump and Dexcom G4 Platinum CGM system into a single device. The t:slim G4 pump displays CGM graphs and trend information with current insulin delivery activity together on the home screen to help make informed treatment decisions.
The t:slim G4 Pump is the newest addition to Tandem’s family of products, which includes the t:slim and t:flex insulin pumps as well. Each pump in the portfolio features a touch-screen with a streamlined, user-friendly interface. This is meant to provide rapid access to frequently used features while eliminating the need for excessive scrolling and button pressing, making these features simple to learn and simple to use. Each Tandem product includes features such as a built-in rechargeable battery and micro-USB port for convenient charging and fast data transfers. They also feature the company’s Micro-Delivery technology that allows for basal delivery every 5 minutes in increments as small as 0.001 unit. The t:slim G4 and t:slim pumps are designed to be very slim and small while still holding up to 300 units of insulin. The t:flex offers similar size benefits while holding up to 480 units of insulin.
“The t:slim G4 Pump combines our intuitive touch-screen interface and bright color display with Dexcom’s CGM technology, giving the diabetes community a powerful new tool to help simplify therapy management,” said Kim Blickenstaff, president and CEO of Tandem Diabetes Care. “CGM integration has been one of the most requested features since the launch of the t:slim pump. The addition of t:slim G4 to our product family allows us to address a wider variety of diabetes management needs through the broadest range of insulin pump options available from one company.”
“The integration of Tandem’s touch-screen interface and Dexcom’s G4 Platinum CGM product could prove to be a powerful combination for people with diabetes,” said Kupper Wintergerst, M.D., pediatric endocrinologist at the University of Louisville and director of the Wendy L. Novak Diabetes Care Center in Louisville, Ky. “It’s great to have another integrated option for my patients, particularly one with an age indication for people as young as 12 years old.”
“We believe the t:slim G4 Pump’s touch screen technology and simple user interface are a great combination for Dexcom G4 Platinum CGM integration,” said Steve Pacelli, executive vice president of strategy and corporate development at Dexcom. “CGM-integration is key to future automated insulin delivery technology, and we are pleased to be working with a company like Tandem who is continuously innovating to make diabetes technology simpler to use for people with diabetes.”
The t:slim G4 Pump is the newest addition to Tandem’s family of products, which includes the t:slim and t:flex insulin pumps as well. Each pump in the portfolio features a touch-screen with a streamlined, user-friendly interface. This is meant to provide rapid access to frequently used features while eliminating the need for excessive scrolling and button pressing, making these features simple to learn and simple to use. Each Tandem product includes features such as a built-in rechargeable battery and micro-USB port for convenient charging and fast data transfers. They also feature the company’s Micro-Delivery technology that allows for basal delivery every 5 minutes in increments as small as 0.001 unit. The t:slim G4 and t:slim pumps are designed to be very slim and small while still holding up to 300 units of insulin. The t:flex offers similar size benefits while holding up to 480 units of insulin.
“The t:slim G4 Pump combines our intuitive touch-screen interface and bright color display with Dexcom’s CGM technology, giving the diabetes community a powerful new tool to help simplify therapy management,” said Kim Blickenstaff, president and CEO of Tandem Diabetes Care. “CGM integration has been one of the most requested features since the launch of the t:slim pump. The addition of t:slim G4 to our product family allows us to address a wider variety of diabetes management needs through the broadest range of insulin pump options available from one company.”
“The integration of Tandem’s touch-screen interface and Dexcom’s G4 Platinum CGM product could prove to be a powerful combination for people with diabetes,” said Kupper Wintergerst, M.D., pediatric endocrinologist at the University of Louisville and director of the Wendy L. Novak Diabetes Care Center in Louisville, Ky. “It’s great to have another integrated option for my patients, particularly one with an age indication for people as young as 12 years old.”
“We believe the t:slim G4 Pump’s touch screen technology and simple user interface are a great combination for Dexcom G4 Platinum CGM integration,” said Steve Pacelli, executive vice president of strategy and corporate development at Dexcom. “CGM-integration is key to future automated insulin delivery technology, and we are pleased to be working with a company like Tandem who is continuously innovating to make diabetes technology simpler to use for people with diabetes.”