08.17.15
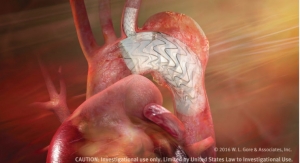
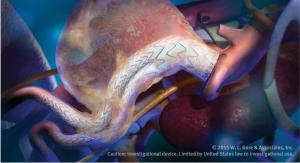
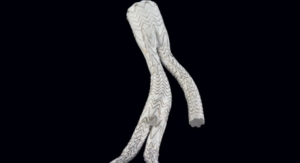
W. L. Gore & Associates' (Gore) Excluder Iliac Branch Endoprosthesis -- a fully engineered system (Gore designed iliac branch and internal iliac components) intended for endovascular treatment of common iliac artery aneurysms or aortoiliac aneurysms -- has been included on the Australian Register of Therapeutic Goods, the company announced.
The first patient procedures in Australia were successfully completed by vascular surgeons Steven Dubenec, M.D., head of the Vascular Surgery Deartment at Royal Prince Alfred Hospital in Sydney, and Jack Loa, M.D., at Macquarie University Hospital, also in Sydney.
This new device — used in conjunction with the Gore Excluder AAA Endoprosthesis components to isolate the common iliac artery from systemic blood flow and preserve blood flow in the external iliac and internal iliac arteries — is built on Gore’s technology platform and designed using the same durable, expanded polytetrafluoroethylene (ePTFE) graft.
“Based on the design of the Gore Excluder device, this system provides a wide treatment range and long-term durability, which is extremely important for optimal clinical success,” said Dubenec. “The pre-cannulated branch and bi-femoral delivery elements of the Gore Excluder Iliac Branch Endoprosthesis support the ease of use of this device.”
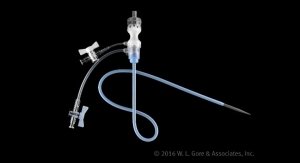
The Gore Excluder Iliac Branch Endoprosthesis system provides a treatment range of 6.5 millimeters to 13.5 millimeters for the internal iliac arteries, and a treatment range of 6.5 millimeters to 25 millimeters for the external iliac arteries. The delivery profile of the loaded catheter allows the use of a 16 Fr introducer sheath for the iliac branch component, and a 12 Fr flexible, reinforced introducer sheath for the internal iliac component.
“This new, fully engineered system builds off the trusted performance of the Gore Excluder AAA Endoprosthesis, providing physicians with a minimally invasive treatment option for iliac repair,” said Ryan Takeuchi, Gore Aortic Business leader. “We have made great strides in advancing our Gore Excluder Device and branch endoprosthesis portfolio and remain committed to making these devices available to more patients across the world.”
The Gore Excluder Iliac Branch Endoprosthesis is part of the family of Gore Excluder Device products.
Gore's products include vascular grafts, endovascular and interventional devices, surgical meshes for hernia repair, soft-tissue reconstruction, staple line reinforcement and sutures for use in vascular, cardiac, and general surgery.
The first patient procedures in Australia were successfully completed by vascular surgeons Steven Dubenec, M.D., head of the Vascular Surgery Deartment at Royal Prince Alfred Hospital in Sydney, and Jack Loa, M.D., at Macquarie University Hospital, also in Sydney.
This new device — used in conjunction with the Gore Excluder AAA Endoprosthesis components to isolate the common iliac artery from systemic blood flow and preserve blood flow in the external iliac and internal iliac arteries — is built on Gore’s technology platform and designed using the same durable, expanded polytetrafluoroethylene (ePTFE) graft.
“Based on the design of the Gore Excluder device, this system provides a wide treatment range and long-term durability, which is extremely important for optimal clinical success,” said Dubenec. “The pre-cannulated branch and bi-femoral delivery elements of the Gore Excluder Iliac Branch Endoprosthesis support the ease of use of this device.”
The Gore Excluder Iliac Branch Endoprosthesis system provides a treatment range of 6.5 millimeters to 13.5 millimeters for the internal iliac arteries, and a treatment range of 6.5 millimeters to 25 millimeters for the external iliac arteries. The delivery profile of the loaded catheter allows the use of a 16 Fr introducer sheath for the iliac branch component, and a 12 Fr flexible, reinforced introducer sheath for the internal iliac component.
“This new, fully engineered system builds off the trusted performance of the Gore Excluder AAA Endoprosthesis, providing physicians with a minimally invasive treatment option for iliac repair,” said Ryan Takeuchi, Gore Aortic Business leader. “We have made great strides in advancing our Gore Excluder Device and branch endoprosthesis portfolio and remain committed to making these devices available to more patients across the world.”
The Gore Excluder Iliac Branch Endoprosthesis is part of the family of Gore Excluder Device products.
Gore's products include vascular grafts, endovascular and interventional devices, surgical meshes for hernia repair, soft-tissue reconstruction, staple line reinforcement and sutures for use in vascular, cardiac, and general surgery.