Officials Direct Flow Medical Inc., reported positive two-year data from the Discover CE mark trial studying the company's Transcatheter Aortic Valve System at the EuroPCR conference in Paris, France. Demonstrating excellent patient outcomes with few complications, the two-year data were presented by Antonio Colombo, M.D., Ph.D., from the Ospedale San Raffaele in Milan, Italy.
According to the company, the Discover trial demonstrated an 80 percent survival rate at 24 months, continuing the positive trend showing 90 percent survival after one year and 99 percent after 30 days. All patients experienced mild or less post-procedural aortic regurgitation (AR), with 85 percent having none or trace AR. Aortic regurgitation is a condition that occurs when the heart's aortic valve doesn't close tightly. Aortic valve regurgitation allows some of the blood that was just pumped out of your heart's main pumping chamber (left ventricle) to leak back into it. The mean gradient remained stable at 12.4 mmHg at 24 months, compared to 12.6 mmHg at 30 days. At the two-year mark, 92 percent of patients had improved by more than one New York Heart Association (NYHA) functional class, improving upon the 83 percent of patients that had improved by more than one NYHA functional class at 30 days. All hemodynamic outcomes were assessed and reported by an independent imaging core laboratory and were sustained over time.
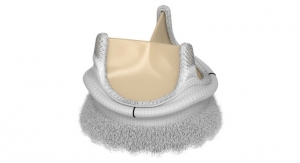
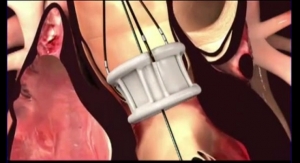
The fully repositionable and retrievable Direct Flow Medical system is indicated to treat patients with severe aortic stenosis who are at extreme surgical risk, while reducing the risk of post-procedural AR, a strong predictor of long-term mortality, according to the company. The system addresses this clinical concern by sealing the annulus (base of a heart valve that supports the valve's leaflets is called the annulus) and enabling complete assessment of hemodynamic performance with in-situ repositioning of the valve after full deployment.
“The 24 month Discover results demonstrate that the initial clinical improvements seen with the Direct Flow Medical valve translate into exceptional long-term outcomes,” said Colombo. “This next generation TAVI (transcather aortic valve implantation) device is the first to report such outstanding results treating an extremely sick patient population.”
The Discover trial is a prospective, multi-center study conducted at nine European sites of 100 patients with severe aortic valve stenosis who required replacement of their native aortic valve but were at extreme risk for open surgical repair.
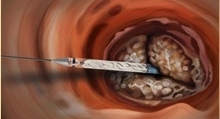
The Direct Flow Medical system also avoids both rapid pacing of the heart during deployment and post-dilatation following placement, minimizing the risk of hemodynamic instability for patients. It includes a distinctive heart valve with a metal-free frame, delivered transfemorally (through the leg) via the same flexible delivery system for all sizes (23 mm, 25 mm, 27 mm and 29 mm).
The Direct Flow Medical system received the CE mark in January 2013. The system is currently available commercially in Europe and enrolling a pivotal investigational device exemption trial in the United States.
Founded in 2004, venture-backed Direct Flow Medical is headquartered in Santa Rosa, Calif., with technology and manufacturing facilities in Lake Forest, Calif.