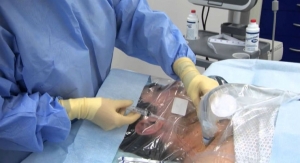
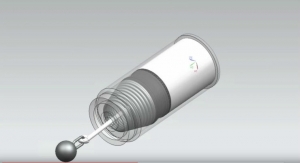
The single-arm study will gauge the safety and efficacy of the Angel Catheter in subjects at high risk of PE, building on positive results gleaned from a European registry and a U.S. feasibility study, BiO2 said. Once completed, BiO2 will seek a prophylactic indication, the first for an inferior vena cava filter.
The catheter offers the acute protection of a retrievable Nitinol IVC filter, permanently attached to a multilumen central venous catheter and can be placed directly at the patient’s bedside without fluoroscopy, according to BiO2. Complete removal of the filter is ensured when the catheter is retrieved — significantly reducing complications that can occur with traditional IVC filters, the company added.
"The pivotal clinical trial is the culmination of years of investigation in this clinical device. The results of this trial will also provide the important clinical data to understand the prophylactic role of this IVC filter and catheter combination. Dr. Victor Tapson, a well-recognized expert in the area of pulmonary embolism is the principal investigator for this trial, in association with a large group of trauma and critical care physicians who feel that positive results of this trial will provide a substantial contribution in the prevention of significant pulmonary embolism events that are associated with increased morbidity and mortality," BiO2 Medical President/Chief Medical Officer Luis Angel.
Founded in 2006, BiO2 Medical has research and development and manufacturing operations in Golden, Colo.