03.10.15
ZOLL Medical Corporation said its Japanese subsidiary, Asahi Kasei Zoll Medical Corp. (AZM), is seeking Japanese approval for its Intravascular Temperature Management (IVTM) System, used for sudden cardiac arrest. The company recently submitted an application to Japan’s Pharmaceuticals and Medical Devices Agency (PMDA).
AZM conducted a clinical study in Japan from January 2013 to February 2014 for the indication of temperature management after resuscitation from cardiac arrest. The application for the new indication was filed based on the results of this study following consultation and advice from the PMDA. The application also includes approval for Zoll’s Quattro and Icy as new catheters for use in temperature management. Currently, only the Cool Line catheter has approval.
“Use of temperature management for sudden cardiac arrest is recommended in numerous national guidelines including the Japan Resuscitation Council Guidelines 2010, which we think will be informative in the PMDA’s consideration of our application,” said James A. Palazzolo, President of Zoll. “In treating resuscitated patients, there is a need for easy-to-use equipment that enables precise cooling and rewarming of core body temperature. By expanding indications for IVTM in Japan, AZM seeks to contribute to advance technology for resuscitation therapy.”
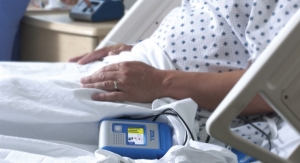
Zoll’s IVTM system provides cooling and warming through a balloon catheter inserted into the venous system. This provides precise control of the core body temperature over surface methods by directly cooling or warming the patient’s blood as it flows through the body. This catheter is connected to the Thermogard XP console, which circulates cold saline solution inside the catheter. Since the saline flows only within the catheter, no fluid is infused into the patient. Zoll offers a variety of triple-lumen catheters with placement options to handle specific patient challenges. The Zoll IVTM system is precise, efficient, and easy to use, and frees up clinical staff to attend to other patient needs. No product has been specifically approved in the United States for cardiac arrest.
Zoll Medical Corporation, an Asahi Kasei Group company, develops and markets medical devices and software solutions that help advance emergency care and save lives, while increasing clinical and operational efficiencies. With products for defibrillation and monitoring, circulation and CPR feedback, data management, fluid resuscitation, and therapeutic temperature management, Zoll develops technologies that help clinicians, EMS and fire professionals, and lay rescuers treat victims needing resuscitation and acute critical care.
The Asahi Kasei Group is a diversified group of companies led by holding company Asahi Kasei Corp., with operations in the chemicals and fibers, homes and construction materials, electronics, and healthcare business sectors. Its healthcare operations include devices and systems for acute critical care, dialysis, therapeutic apheresis, transfusion, and manufacture of biotherapeutics, as well as pharmaceuticals, diagnostic reagents, and nutritional products. With more than 29,000 employees worldwide, the Asahi Kasei Group serves customers in more than 100 countries.
AZM conducted a clinical study in Japan from January 2013 to February 2014 for the indication of temperature management after resuscitation from cardiac arrest. The application for the new indication was filed based on the results of this study following consultation and advice from the PMDA. The application also includes approval for Zoll’s Quattro and Icy as new catheters for use in temperature management. Currently, only the Cool Line catheter has approval.
“Use of temperature management for sudden cardiac arrest is recommended in numerous national guidelines including the Japan Resuscitation Council Guidelines 2010, which we think will be informative in the PMDA’s consideration of our application,” said James A. Palazzolo, President of Zoll. “In treating resuscitated patients, there is a need for easy-to-use equipment that enables precise cooling and rewarming of core body temperature. By expanding indications for IVTM in Japan, AZM seeks to contribute to advance technology for resuscitation therapy.”
Zoll’s IVTM system provides cooling and warming through a balloon catheter inserted into the venous system. This provides precise control of the core body temperature over surface methods by directly cooling or warming the patient’s blood as it flows through the body. This catheter is connected to the Thermogard XP console, which circulates cold saline solution inside the catheter. Since the saline flows only within the catheter, no fluid is infused into the patient. Zoll offers a variety of triple-lumen catheters with placement options to handle specific patient challenges. The Zoll IVTM system is precise, efficient, and easy to use, and frees up clinical staff to attend to other patient needs. No product has been specifically approved in the United States for cardiac arrest.
Zoll Medical Corporation, an Asahi Kasei Group company, develops and markets medical devices and software solutions that help advance emergency care and save lives, while increasing clinical and operational efficiencies. With products for defibrillation and monitoring, circulation and CPR feedback, data management, fluid resuscitation, and therapeutic temperature management, Zoll develops technologies that help clinicians, EMS and fire professionals, and lay rescuers treat victims needing resuscitation and acute critical care.
The Asahi Kasei Group is a diversified group of companies led by holding company Asahi Kasei Corp., with operations in the chemicals and fibers, homes and construction materials, electronics, and healthcare business sectors. Its healthcare operations include devices and systems for acute critical care, dialysis, therapeutic apheresis, transfusion, and manufacture of biotherapeutics, as well as pharmaceuticals, diagnostic reagents, and nutritional products. With more than 29,000 employees worldwide, the Asahi Kasei Group serves customers in more than 100 countries.