03.05.15
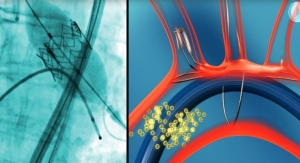
Essential Medical,Inc. has successfully completed First in Man studies for its Manta Large Bore Vascular Closure device, a product designed to seal 18F large bore femoral punctures.
18F Manta devices were safely and effectively deployed in five patients in this initial series, achieving immediate or rapid hemostasis at the large bore puncture site with excellent clinical, angiographic and hemodynamic (Duplex) success in all patients, the company reported. Patients were discharged the following day and there was no evidence of hematoma in any patient. The Manta devices were deployed in less than one minute in each patient.
Manta closes femoral arterial punctures made during cardiac catheterization procedures such as transcatheter aortic valve replacement, balloon aortic valvuloplasty, and the endovascular treatment of abdominal aortic aneurysms. Manta can be deployed in less than 60 seconds and utilizes sealing components similar to the company's other closure device, X-Seal. The potential worldwide market for vascular closure products is approximately $1 billion, company officials claim.
"In interventional cardiology it's critical to define the problem that the patient is facing, solve the problem and use the best clinical science available to confirm beneficial outcomes. We're doing that with Manta and it promises to be a transformative technology for providing percutaneous access for large bore therapeutic devices," said Gary Roubin, M.D., Ph.D., an interventional cardiologist who performed the first two cases.
"This device fills a critical unmet need for endovascular procedures in being able to quickly and reliably close large bore catheter channels. This ability, done simply and safely is a major step to full percutaneous procedures in both the cardiac and peripheral markets," said Lawrence Garcia, M.D., chief of Interventional Cardiology at St. Elizabeth's Medical Center in Boston, Mass., who also participated in the study.
Large bore femoral access has been associated with significant morbidity including bleeding complications, the need for transfusions, increased hospital costs, patient discomfort and disability. "Our team is thrilled to bring forward a device technology that can manage challenging 14F and 18F closures," said Greg Walters, Essential Medical CEO. "We look forward to initiating a CE-marking study in early 2015."
Essential Medical is a privately held medical device company that develops and commercializes vascular closure devices for femoral punctures after diagnostic and interventional cardiac catheterization procedures. The Malvern, Pa.-based company was founded in 2010 by Walters and Roubin.
18F Manta devices were safely and effectively deployed in five patients in this initial series, achieving immediate or rapid hemostasis at the large bore puncture site with excellent clinical, angiographic and hemodynamic (Duplex) success in all patients, the company reported. Patients were discharged the following day and there was no evidence of hematoma in any patient. The Manta devices were deployed in less than one minute in each patient.
Manta closes femoral arterial punctures made during cardiac catheterization procedures such as transcatheter aortic valve replacement, balloon aortic valvuloplasty, and the endovascular treatment of abdominal aortic aneurysms. Manta can be deployed in less than 60 seconds and utilizes sealing components similar to the company's other closure device, X-Seal. The potential worldwide market for vascular closure products is approximately $1 billion, company officials claim.
"In interventional cardiology it's critical to define the problem that the patient is facing, solve the problem and use the best clinical science available to confirm beneficial outcomes. We're doing that with Manta and it promises to be a transformative technology for providing percutaneous access for large bore therapeutic devices," said Gary Roubin, M.D., Ph.D., an interventional cardiologist who performed the first two cases.
"This device fills a critical unmet need for endovascular procedures in being able to quickly and reliably close large bore catheter channels. This ability, done simply and safely is a major step to full percutaneous procedures in both the cardiac and peripheral markets," said Lawrence Garcia, M.D., chief of Interventional Cardiology at St. Elizabeth's Medical Center in Boston, Mass., who also participated in the study.
Large bore femoral access has been associated with significant morbidity including bleeding complications, the need for transfusions, increased hospital costs, patient discomfort and disability. "Our team is thrilled to bring forward a device technology that can manage challenging 14F and 18F closures," said Greg Walters, Essential Medical CEO. "We look forward to initiating a CE-marking study in early 2015."
Essential Medical is a privately held medical device company that develops and commercializes vascular closure devices for femoral punctures after diagnostic and interventional cardiac catheterization procedures. The Malvern, Pa.-based company was founded in 2010 by Walters and Roubin.