02.25.15
Intact Vascular has finished enrolling patients in its Tack Optimized Balloon Angioplasty - Below the Knee Study.
The multi-center pilot study is designed to evaluate the safety and performance of the Tack Endovascular System in subjects with Critical Limb Ischemia (CLI) due to vascular disease below the knee. Thirty-five subjects were enrolled in the study, which targeted diseased tibial vessels for treatment.
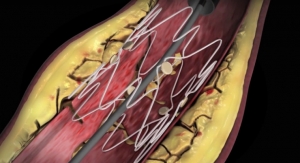
CLI is an advanced stage of peripheral artery disease in which patients suffer from inadequate blood flow to the lower leg and foot. If left untreated, this may lead to pain, gangrene and tissue loss which can result in amputation. Balloon angioplasty is the most common procedure performed to restore blood flow in these blocked arteries. However, post-angioplasty dissection often occurs at the treatment site, which increases the risk of thrombotic occlusion.
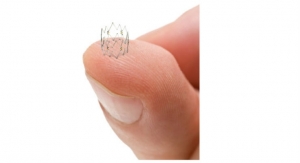
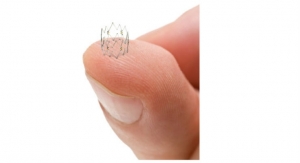
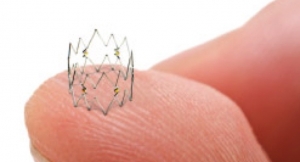
The Tack device is designed to repair tissue tears at the site of post-angioplasty dissection. The Tack Endovascular System has been designed to leave minimal foreign material in the artery; apply only the minimum outward force required to repair tissue tears within the lumen; and allow “spot” treatment only where needed. The system is composed of a delivery catheter containing three self-expanding nitinol tacks.
“Critical limb ischemia is a devastating and costly disease that demands new, innovative treatments. Completion of the TOBA-BTK trial is an important step toward developing an important new tool that could ultimately save the limbs of patients suffering from CLI,” said assistant professor Christian Wissgott, M.D., deputy chief, Institute of Diagnostic and Interventional Radiology, Westkuestenklinikum Heide, Germany. Wissgott also was a leading enroller in the trial.
“Our best method for salvaging threatened limbs is to revascularize them using balloon angioplasty, but the current technique has limitations. The Tack Endovascular System has the potential to substantially improve outcomes for CLI patients by optimizing angioplasty,” said Peter Schneider, M.D., Intact Vascular chief medical officer and chief, Vascular Therapy at Kaiser Foundation Hospital in Honolulu, Hawaii.
Based in Wayne, Pa., Intact Vascular is a privately held medical device company that develops minimally invasive peripheral vascular products. The Tack Endovascular System is designed to optimize balloon angioplasty results in the treatment of peripheral artery disease.
The multi-center pilot study is designed to evaluate the safety and performance of the Tack Endovascular System in subjects with Critical Limb Ischemia (CLI) due to vascular disease below the knee. Thirty-five subjects were enrolled in the study, which targeted diseased tibial vessels for treatment.
CLI is an advanced stage of peripheral artery disease in which patients suffer from inadequate blood flow to the lower leg and foot. If left untreated, this may lead to pain, gangrene and tissue loss which can result in amputation. Balloon angioplasty is the most common procedure performed to restore blood flow in these blocked arteries. However, post-angioplasty dissection often occurs at the treatment site, which increases the risk of thrombotic occlusion.
The Tack device is designed to repair tissue tears at the site of post-angioplasty dissection. The Tack Endovascular System has been designed to leave minimal foreign material in the artery; apply only the minimum outward force required to repair tissue tears within the lumen; and allow “spot” treatment only where needed. The system is composed of a delivery catheter containing three self-expanding nitinol tacks.
“Critical limb ischemia is a devastating and costly disease that demands new, innovative treatments. Completion of the TOBA-BTK trial is an important step toward developing an important new tool that could ultimately save the limbs of patients suffering from CLI,” said assistant professor Christian Wissgott, M.D., deputy chief, Institute of Diagnostic and Interventional Radiology, Westkuestenklinikum Heide, Germany. Wissgott also was a leading enroller in the trial.
“Our best method for salvaging threatened limbs is to revascularize them using balloon angioplasty, but the current technique has limitations. The Tack Endovascular System has the potential to substantially improve outcomes for CLI patients by optimizing angioplasty,” said Peter Schneider, M.D., Intact Vascular chief medical officer and chief, Vascular Therapy at Kaiser Foundation Hospital in Honolulu, Hawaii.
Based in Wayne, Pa., Intact Vascular is a privately held medical device company that develops minimally invasive peripheral vascular products. The Tack Endovascular System is designed to optimize balloon angioplasty results in the treatment of peripheral artery disease.