12.11.14
Bioengineers from ETH Zurich University's Department of Biosystems Science and Engineering in Switzerland have created a prototype implantable molecular device which regulates the blood pH levels through a closed loop pH sensing and insulin production mechanism.
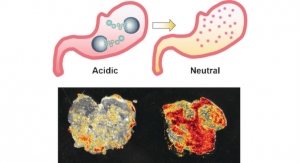
Normal metabolic function requires acid levels in the body to be tightly regulated. If the pH level of the blood strays too far from a narrow band of healthy values there are severe consequences in the form of impaired organ function, seizures, shock, and possibly death.
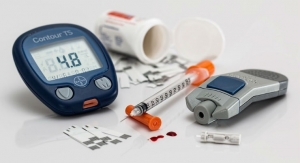
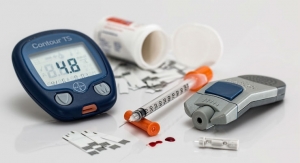
pH regulation particularly is problematic in people with type 1 diabetes. As their bodies do not produce their own insulin, their cells metabolize fat reserves. This in turn produces beta-hydroxybutyrate in the liver, an acid which serves as an energy source when blood glucose is low. If the liver produces too much beta-hydroxybutyrate the blood’s pH value can drop to dangerous levels, a condition known as acidosis.
The molecular device was designed from various genes and proteins and comprises a pH sensing element and a gene feedback mechanism which produces insulin. The pH sensor monitors the blood’s acidity with a high degree of precision and reacts to minor deviations from the ideal pH value. If the pH value dips below 7.35, a critically low value for people with type 1 diabetes, the pH sensor triggers insulin production. The insulin causes the cells to metabolise glucose instead of fat reserves and returns the blood pH to a healthy level.
To date the researchers have tested their device in mice with type 1 diabetes and related acidosis. Effective insulin production was observed in mice with the implanted capsules, having hormone levels comparable to that of healthy mice. The implant was able to successfully counteract the effects of significant changes in blood sugar levels.
“Applications for humans are conceivable based on this prototype, but they are yet to be developed,” said Martin Fussenegger, a biotechnology/bioengineering professor. “We wanted to create a prototype first to see whether molecular prostheses could even be used for such fine adjustments to metabolic processes.”
While the present system is an initial proof of concept, the team has recognized possible applications for human implants, though it would require significant time and resources to take to market -- resources the university does not currently have without a collaborative industrial partner. The system design and test results have been published in the journal Molecular Cell.
Researchers in Fussenegger’s group have already made headlines several times with similar synthetic networks. For instance, they developed an implant with genes that could be activated with blue light, thereby producing GLP-1, which regulates insulin production. They also put together a network that eliminates metabolic syndrome, a process set in motion by an authorised blood-pressure medicine. All of these networks respond to a signal and produce a hormonally active substance. The special thing about the new feedback mechanism, however, is that the body itself produces the signal, which is then detected by a sensor that triggers a fine-tuned therapeutic reaction.
Three groups from the D-BSSE worked on the present project. Fussenegger’s group developed the genetic network; Professor of Biosystems Engineering Andreas Hierlemann and his team tested the acidity sensor with the aid of microfluidic platforms; and Jörg Stelling, a professor of computational systems biology, modeled it in order to estimate the dynamics of the insulin production.
Normal metabolic function requires acid levels in the body to be tightly regulated. If the pH level of the blood strays too far from a narrow band of healthy values there are severe consequences in the form of impaired organ function, seizures, shock, and possibly death.
pH regulation particularly is problematic in people with type 1 diabetes. As their bodies do not produce their own insulin, their cells metabolize fat reserves. This in turn produces beta-hydroxybutyrate in the liver, an acid which serves as an energy source when blood glucose is low. If the liver produces too much beta-hydroxybutyrate the blood’s pH value can drop to dangerous levels, a condition known as acidosis.
The molecular device was designed from various genes and proteins and comprises a pH sensing element and a gene feedback mechanism which produces insulin. The pH sensor monitors the blood’s acidity with a high degree of precision and reacts to minor deviations from the ideal pH value. If the pH value dips below 7.35, a critically low value for people with type 1 diabetes, the pH sensor triggers insulin production. The insulin causes the cells to metabolise glucose instead of fat reserves and returns the blood pH to a healthy level.
To date the researchers have tested their device in mice with type 1 diabetes and related acidosis. Effective insulin production was observed in mice with the implanted capsules, having hormone levels comparable to that of healthy mice. The implant was able to successfully counteract the effects of significant changes in blood sugar levels.
“Applications for humans are conceivable based on this prototype, but they are yet to be developed,” said Martin Fussenegger, a biotechnology/bioengineering professor. “We wanted to create a prototype first to see whether molecular prostheses could even be used for such fine adjustments to metabolic processes.”
While the present system is an initial proof of concept, the team has recognized possible applications for human implants, though it would require significant time and resources to take to market -- resources the university does not currently have without a collaborative industrial partner. The system design and test results have been published in the journal Molecular Cell.
Researchers in Fussenegger’s group have already made headlines several times with similar synthetic networks. For instance, they developed an implant with genes that could be activated with blue light, thereby producing GLP-1, which regulates insulin production. They also put together a network that eliminates metabolic syndrome, a process set in motion by an authorised blood-pressure medicine. All of these networks respond to a signal and produce a hormonally active substance. The special thing about the new feedback mechanism, however, is that the body itself produces the signal, which is then detected by a sensor that triggers a fine-tuned therapeutic reaction.
Three groups from the D-BSSE worked on the present project. Fussenegger’s group developed the genetic network; Professor of Biosystems Engineering Andreas Hierlemann and his team tested the acidity sensor with the aid of microfluidic platforms; and Jörg Stelling, a professor of computational systems biology, modeled it in order to estimate the dynamics of the insulin production.