12.10.14
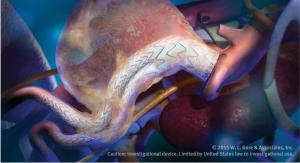
The Viabahn Endoprosthesis from W. L. Gore & Associates Inc. received CE mark approval in Europe. The technology is designed to improve blood flow in symptomatic obstruction of peripheral veins, excluding the venae cavae and pulmonary veins.
“The GoreViabahn Endoprosthesis is a welcome addition to options available for treating stenosis in the dialysis access circuit,” said Peter Riley, M.D., consultant interventional radiologist at the Queen Elizabeth Hospital in Birmingham, the United Kingdom. “It has a proven track record in maintaining patency in central chest vein stenosis as confirmed by our recently published single-center study. It is extremely flexible, allowing deployment across anatomical sites of movement and flexion. The Gore REVISE clinical study indicates improved patency compared to PTA (percutaneous transluminal angioplasty) in the graft to vein anastamosis and has demonstrated similar outcomes to draining veins, particularly at the cephalic arch, in my practice.”
PTA is a type of balloon angioplasty in which the catheter is inserted through the skin and through the lumen of the vessel to the site of the narrowing.
According to Gore officials, in the REVISE study, the company’s Viabahn device group “demonstrated statistical superiority of target lesion primary patency” when compared to PTA (p = 0.008).
According to the company, “with a long history of iliac and superficial femoral artery use, the Viabahn device is the lowest profile, most flexible, self-expanding, small-diameter, endoprosthesis available.”
The device is made with a durable, reinforced, biocompatible, expanded polytetrafluoroethylene (ePTFE) liner and attached to an external nitinol stent structure. The ePTFE luminal surface incorporates the CBAS Heparin Surface made by Carmeda AB, a subsidiary of Gore. The coating is designed to actively reduce thrombus formation—or clotting—on blood-contacting medical devices.
“We are pleased to expand the Gore Viabahn Endoprosthesis to hemodialysis access in Europe,” said Ray Swinney, business unit leader for Gore’s Peripheral Interventional division. “We are committed to exceeding the expectations of vascular surgeons, interventional radiologists, and interventional nephrologists who have been seeking a better solution for their dialysis access patients.”
Flagstaff, Ariz.-based Gore Medical makes vascular grafts, endovascular and interventional devices, surgical meshes for hernia repair, soft tissue reconstruction, staple line reinforcement and sutures for use in vascular, cardiac, and general surgery.
“The GoreViabahn Endoprosthesis is a welcome addition to options available for treating stenosis in the dialysis access circuit,” said Peter Riley, M.D., consultant interventional radiologist at the Queen Elizabeth Hospital in Birmingham, the United Kingdom. “It has a proven track record in maintaining patency in central chest vein stenosis as confirmed by our recently published single-center study. It is extremely flexible, allowing deployment across anatomical sites of movement and flexion. The Gore REVISE clinical study indicates improved patency compared to PTA (percutaneous transluminal angioplasty) in the graft to vein anastamosis and has demonstrated similar outcomes to draining veins, particularly at the cephalic arch, in my practice.”
PTA is a type of balloon angioplasty in which the catheter is inserted through the skin and through the lumen of the vessel to the site of the narrowing.
According to Gore officials, in the REVISE study, the company’s Viabahn device group “demonstrated statistical superiority of target lesion primary patency” when compared to PTA (p = 0.008).
According to the company, “with a long history of iliac and superficial femoral artery use, the Viabahn device is the lowest profile, most flexible, self-expanding, small-diameter, endoprosthesis available.”
The device is made with a durable, reinforced, biocompatible, expanded polytetrafluoroethylene (ePTFE) liner and attached to an external nitinol stent structure. The ePTFE luminal surface incorporates the CBAS Heparin Surface made by Carmeda AB, a subsidiary of Gore. The coating is designed to actively reduce thrombus formation—or clotting—on blood-contacting medical devices.
“We are pleased to expand the Gore Viabahn Endoprosthesis to hemodialysis access in Europe,” said Ray Swinney, business unit leader for Gore’s Peripheral Interventional division. “We are committed to exceeding the expectations of vascular surgeons, interventional radiologists, and interventional nephrologists who have been seeking a better solution for their dialysis access patients.”
Flagstaff, Ariz.-based Gore Medical makes vascular grafts, endovascular and interventional devices, surgical meshes for hernia repair, soft tissue reconstruction, staple line reinforcement and sutures for use in vascular, cardiac, and general surgery.