11.12.14
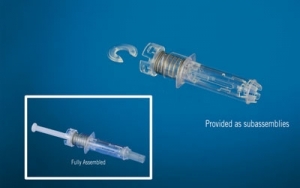
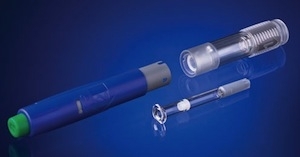
West Pharmaceutical Services Inc., a developer of devices for injectable drug administration, reported that its NovaGuard SA (staked-needle automatic) safety system has received 510(k) clearance from the U.S. Food and Drug Administration. The NovaGuard SA safety system helps reduce the risk of needle-stick injuries by shielding the exposed needle of a pre-filled syringe after use, according to the company.
The World Health Organization (WHO) estimates that each year healthcare workers incur two million accidental needlestick injuries that may result in infections with hepatitis B and C, HIV and other diseases. Acknowledging the public health threat, WHO, the European Union, the Occupational Safety and Health Administration and other leading health organizations have developed guidelines focused on needle safety and recommending the use of safety-engineered devices to reduce sharps injuries.
"With increased global emphasis on preventing the spread of bloodborne infectious diseases, there is a need to develop and implement safer systems and practices in healthcare facilities worldwide. This includes adopting devices that can help reduce the risk of infection caused by needlestick," said Graham Reynolds, vice president, marketing and innovation, West Pharmaceutical Services. "West has deep expertise in needlestick prevention and a portfolio of offerings that enable safe and effective drug administration. It's our hope that the NovaGuard SA safety system is used as a cost-effective solution to help improve needle safety."
The NovaGuard SA safety system helps protect patients and healthcare workers by shielding a syringe's exposed needle once activated. The system is suitable for prefilled ISO standard glass syringes and provides a tamper-evident needle shield feature and a safety mechanism, which helps to prevent pre-activation of the system. It is part of West's NovaGuard family of syringe safety technology offerings.
West has increased its manufacturing capacity at its facility in France, where more than 1.5 billion similar safety systems already have been produced. The NovaGuard SA safety system could be available for all geographic markets and commercial supply. Currently, the NovaGuard SA safety system is commercialized with one marketed drug outside of the U.S., and is being evaluated by multiple West partners.
Based in Exton, Pa., West helps its healthcare partners from concept to the patient, designing and manufacturing packaging, diagnostic and delivery systems. West's 2013 sales reflect the daily use of approximately 100 million of its components and devices around the world.
The World Health Organization (WHO) estimates that each year healthcare workers incur two million accidental needlestick injuries that may result in infections with hepatitis B and C, HIV and other diseases. Acknowledging the public health threat, WHO, the European Union, the Occupational Safety and Health Administration and other leading health organizations have developed guidelines focused on needle safety and recommending the use of safety-engineered devices to reduce sharps injuries.
"With increased global emphasis on preventing the spread of bloodborne infectious diseases, there is a need to develop and implement safer systems and practices in healthcare facilities worldwide. This includes adopting devices that can help reduce the risk of infection caused by needlestick," said Graham Reynolds, vice president, marketing and innovation, West Pharmaceutical Services. "West has deep expertise in needlestick prevention and a portfolio of offerings that enable safe and effective drug administration. It's our hope that the NovaGuard SA safety system is used as a cost-effective solution to help improve needle safety."
The NovaGuard SA safety system helps protect patients and healthcare workers by shielding a syringe's exposed needle once activated. The system is suitable for prefilled ISO standard glass syringes and provides a tamper-evident needle shield feature and a safety mechanism, which helps to prevent pre-activation of the system. It is part of West's NovaGuard family of syringe safety technology offerings.
West has increased its manufacturing capacity at its facility in France, where more than 1.5 billion similar safety systems already have been produced. The NovaGuard SA safety system could be available for all geographic markets and commercial supply. Currently, the NovaGuard SA safety system is commercialized with one marketed drug outside of the U.S., and is being evaluated by multiple West partners.
Based in Exton, Pa., West helps its healthcare partners from concept to the patient, designing and manufacturing packaging, diagnostic and delivery systems. West's 2013 sales reflect the daily use of approximately 100 million of its components and devices around the world.