11.10.14
Biotronik SE & Co. KG has agreed to pay $4.9 million to the federal government to resolve allegations the firm paid kickbacks to doctors in Nevada and Arizona to use its products.
The settlement was sparked by a whistleblower lawsuit known as a qui tam, which the U.S. Department of Justice (DOJ) agreed to join in federal civil court.
According to the settlement, Biotronik continues to deny the allegations, echoing statements executives told The Oregonian last year. "We are really clean when it comes to our relationships with physicians," Biotronik's U.S. president, Jake Langer, said last summer.
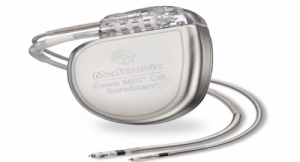
The settlement resolves allegations that Biotronik, through the payment of kickbacks to physicians, caused hospitals and ambulatory surgery centers to submit false claims to Medicare and Medicaid for the implantation of Biotronik pacemakers, defibrillators and cardiac resynchronization therapy devices. Prosecutors claim Biotronik allegedly induced electrophysiologists and cardiologists practicing in Nevada and Arizona to continue using the company's devices, or to convert to its devices, by paying the implanting physician in the form of repeated meals at expensive restaurants and inflated payments for membership on a physician advisory board.
“When medical device manufacturers make improper payments to physicians, they encourage medical decision-making based on financial gain rather than the best interests of patients,” said Acting Assistant Attorney General Joyce R. Branda for the Justice Department’s Civil Division. “The resolution demonstrates the Department of Justice’s continuing commitment to ensuring that beneficiaries of federal health care programs receive appropriate medical care.”
The lawsuit was initially filed by a former Biotronik employee, Brian Sant, who will receive about $840,000 under the federal settlement.
According to DOJ, the investigation leading to the settlement was assisted by the DOJ, the Office of the Inspector General of the Department of Health and Human Services, and the Federal Bureau of Investigation.
Last year, the Oregon Department of Justice setled a case with two Salem, Ore., doctors accusing them of "concealing" from patients payments that created a potential "incentive" to use Biotronik implants, part of a training program that put a sales representative into the operating room.
Biotronik's Langer denied wrongdoing and called the state's case unfair and detrimental to good health care, The Oregonian reported.
Biotronik is based in Berlin, Germany, and has a global workforce of more than 5,600 employees. Its U.S. headquarters is located in Lake Oswego, Ore.
The settlement was sparked by a whistleblower lawsuit known as a qui tam, which the U.S. Department of Justice (DOJ) agreed to join in federal civil court.
According to the settlement, Biotronik continues to deny the allegations, echoing statements executives told The Oregonian last year. "We are really clean when it comes to our relationships with physicians," Biotronik's U.S. president, Jake Langer, said last summer.
The settlement resolves allegations that Biotronik, through the payment of kickbacks to physicians, caused hospitals and ambulatory surgery centers to submit false claims to Medicare and Medicaid for the implantation of Biotronik pacemakers, defibrillators and cardiac resynchronization therapy devices. Prosecutors claim Biotronik allegedly induced electrophysiologists and cardiologists practicing in Nevada and Arizona to continue using the company's devices, or to convert to its devices, by paying the implanting physician in the form of repeated meals at expensive restaurants and inflated payments for membership on a physician advisory board.
“When medical device manufacturers make improper payments to physicians, they encourage medical decision-making based on financial gain rather than the best interests of patients,” said Acting Assistant Attorney General Joyce R. Branda for the Justice Department’s Civil Division. “The resolution demonstrates the Department of Justice’s continuing commitment to ensuring that beneficiaries of federal health care programs receive appropriate medical care.”
The lawsuit was initially filed by a former Biotronik employee, Brian Sant, who will receive about $840,000 under the federal settlement.
According to DOJ, the investigation leading to the settlement was assisted by the DOJ, the Office of the Inspector General of the Department of Health and Human Services, and the Federal Bureau of Investigation.
Last year, the Oregon Department of Justice setled a case with two Salem, Ore., doctors accusing them of "concealing" from patients payments that created a potential "incentive" to use Biotronik implants, part of a training program that put a sales representative into the operating room.
Biotronik's Langer denied wrongdoing and called the state's case unfair and detrimental to good health care, The Oregonian reported.
Biotronik is based in Berlin, Germany, and has a global workforce of more than 5,600 employees. Its U.S. headquarters is located in Lake Oswego, Ore.