11.07.14
Guided Therapeutics Inc. has received approval from the Federal Commission for Protection against Health Risks to sell its LuViva Advanced Cervical Scan in Mexico.
“Approval to sell LuViva in Mexico is a significant milestone for the company and opens up the second-largest market for medical products in Latin America,” said Gene Cartwright, CEO of Guided Therapeutics. “We will be working with Dantien5 Medical, our distributor in Mexico, to promote LuViva for the detection of cervical cancer, a tremendous unmet need in this part of the world.”
Cervical cancer is the leading cause of cancer in women of reproductive age in Mexico, and remains the second leading cause of cancer deaths in women. Each year, more than 13,900 cases of cervical cancer are diagnosed in Mexico, and more than 4,700 women die from the disease. The total population at risk for developing cervical cancer is nearly 36 million (ages 15-55), and cervical cancer remains a national health priority.
“Cervical cancer is a large problem in Mexico with limited access to health care which contributes to a high national mortality rate. As a result, we are actively working on practical evaluations to expedite availability of this early detection technology to Mexican women,” said Carlos Lugo, Director of Dantien5 Medical. “After a recent meeting with the principal investigator at a key facility, we expect the evaluation to begin shortly.”
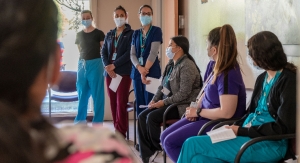
LuViva recently was introduced to several hundred Mexican gynecologists in Mexico City.
Guided Therapeutics describes the LuViva product as a technologically advanced diagnostic device that scans the cervix with light and uses spectroscopy to measure how light interacts with the cervical tissue. Spectroscopy identifies chemical and structural indicators of precancer that may be below the surface of the cervix or misdiagnosed as benign. This technique is called biophotonics. Unlike Pap, HPV tests or biopsies, LuViva does not require laboratory analysis or a tissue sample, and is designed to provide results immediately, which eliminates costly, painful and unnecessary testing. LuViva is designed for use with women who have undergone initial screening and are called back for follow up with a colposcopy examination, which in many cases, involves taking a biopsy of the cervix. The device is used in conjunction with the LuViva Cervical Guide single-use patient interface and calibration disposable. In a multi-center clinical trial, with women at risk for cervical disease, the technology was able to detect cervical cancer up to two years earlier than conventional modalities, according to published reports.
Guided Therapeutics Inc. makes a rapid and painless testing platform based on its patented biophotonic technology that utilizes light for the early detection of disease at the cellular level. Guided Therapeutics is also developing a non-invasive test for the early detection of esophageal cancer using the technology platform. The company is based in Norcross, Ga.
“Approval to sell LuViva in Mexico is a significant milestone for the company and opens up the second-largest market for medical products in Latin America,” said Gene Cartwright, CEO of Guided Therapeutics. “We will be working with Dantien5 Medical, our distributor in Mexico, to promote LuViva for the detection of cervical cancer, a tremendous unmet need in this part of the world.”
Cervical cancer is the leading cause of cancer in women of reproductive age in Mexico, and remains the second leading cause of cancer deaths in women. Each year, more than 13,900 cases of cervical cancer are diagnosed in Mexico, and more than 4,700 women die from the disease. The total population at risk for developing cervical cancer is nearly 36 million (ages 15-55), and cervical cancer remains a national health priority.
“Cervical cancer is a large problem in Mexico with limited access to health care which contributes to a high national mortality rate. As a result, we are actively working on practical evaluations to expedite availability of this early detection technology to Mexican women,” said Carlos Lugo, Director of Dantien5 Medical. “After a recent meeting with the principal investigator at a key facility, we expect the evaluation to begin shortly.”
LuViva recently was introduced to several hundred Mexican gynecologists in Mexico City.
Guided Therapeutics describes the LuViva product as a technologically advanced diagnostic device that scans the cervix with light and uses spectroscopy to measure how light interacts with the cervical tissue. Spectroscopy identifies chemical and structural indicators of precancer that may be below the surface of the cervix or misdiagnosed as benign. This technique is called biophotonics. Unlike Pap, HPV tests or biopsies, LuViva does not require laboratory analysis or a tissue sample, and is designed to provide results immediately, which eliminates costly, painful and unnecessary testing. LuViva is designed for use with women who have undergone initial screening and are called back for follow up with a colposcopy examination, which in many cases, involves taking a biopsy of the cervix. The device is used in conjunction with the LuViva Cervical Guide single-use patient interface and calibration disposable. In a multi-center clinical trial, with women at risk for cervical disease, the technology was able to detect cervical cancer up to two years earlier than conventional modalities, according to published reports.
Guided Therapeutics Inc. makes a rapid and painless testing platform based on its patented biophotonic technology that utilizes light for the early detection of disease at the cellular level. Guided Therapeutics is also developing a non-invasive test for the early detection of esophageal cancer using the technology platform. The company is based in Norcross, Ga.