11.07.14
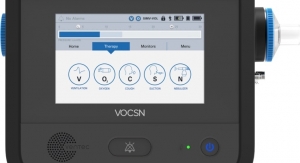
Solna, Sweden-based Aerocrine AB has earned U.S. Food and Drug Administration 510(k) clearance for the next generation of its fractional exhaled nitric oxide (FeNO) monitoring device, Niox Vero. The device is an update of the previous version called Niox Mino.
“The portability and patient-centered improvements provided by Niox Vero will make it more practical for specialists as well as primary care physicians to make FeNO testing a routine part of identifying and managing allergic airway inflammation in patients with diagnosed or suspected asthma,” said CEO Scott.
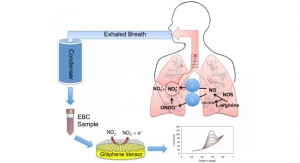
NIOX VERO is a small, point-of-care device that provides a patient’s FeNO score in about one minute after a 10-second exhalation. The device improves on the previous generation Niox Mino by including a rechargeable battery for portability within the clinical setting and an integrated visual display to help patients complete a successful exhalation. Aerocrine’s Niox Mino and now Niox Vero are the only FeNO measurement devices cleared by the FDA and commercially available in the United States.
“FeNO testing is an important addition to clinicians’ tool set because it is the only way to objectively identify and manage allergic airway inflammation, which is the leading cause of asthma symptoms,” said Kathleen Rickard, M.D., chief medical officer at Aerocrine. “Routine FeNO testing helps clinicians find hidden and under-treated allergic airway inflammation, which has been shown to help reduce asthma exacerbations by up to 50 percent compared to usual care.”
Aerocrine makes devices for the treatment of inflammatory airway diseases.
“The portability and patient-centered improvements provided by Niox Vero will make it more practical for specialists as well as primary care physicians to make FeNO testing a routine part of identifying and managing allergic airway inflammation in patients with diagnosed or suspected asthma,” said CEO Scott.
NIOX VERO is a small, point-of-care device that provides a patient’s FeNO score in about one minute after a 10-second exhalation. The device improves on the previous generation Niox Mino by including a rechargeable battery for portability within the clinical setting and an integrated visual display to help patients complete a successful exhalation. Aerocrine’s Niox Mino and now Niox Vero are the only FeNO measurement devices cleared by the FDA and commercially available in the United States.
“FeNO testing is an important addition to clinicians’ tool set because it is the only way to objectively identify and manage allergic airway inflammation, which is the leading cause of asthma symptoms,” said Kathleen Rickard, M.D., chief medical officer at Aerocrine. “Routine FeNO testing helps clinicians find hidden and under-treated allergic airway inflammation, which has been shown to help reduce asthma exacerbations by up to 50 percent compared to usual care.”
Aerocrine makes devices for the treatment of inflammatory airway diseases.