10.16.14
Itamar Medical Ltd. has announced that Nihon Kohden, one of Japan's largest medical device manufacturers, will distribute the EndoPAT device as its leading product for endothelial function assessment, and will market the EndoPAT to hospitals and to more than 100,000 general Japanese practitioners.
The exclusive distribution agreement was reached after Nihon Kohden concluded that testing with Itamar Medical's EndoPAT device provides unique and important indications in assessing the risk of cardiovascular events. The agreement comes one year after Itamar Medical's subsidiary launched operations in Japan.
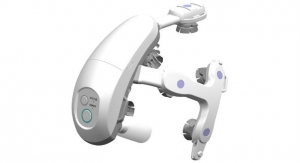
The EndoPAT device is used in non-invasive assessment of arterial health through the assessment of endothelial function (the inner lining of blood vessels), which regulates vasodilation and vasoconstriction. The test takes 15 minutes. A thimble-like sensor is placed on a patient finger on each hand and a blood pressure cuff temporarily stops blood flow to the arm to measure the arterial reaction and expansion once blood flow resumes. Unlike ultrasound-based tests, the EndoPAT device offers ease of use and accurate information that is not reliant on the competency of the testing staff and doctor who interprets the results.
"Nihon Kohden is the ideal partner for distribution of EndoPAT in Japan due to its stature among cardiologists and general practitioners," Itamar Medical vice president of Sales Eldad Singer noted. "The agreement, signed for three years, may be extended. It stipulates aggressive sales and growth targets as a condition for exclusivity."
Over the past year, Itamar Medical has diversified beyond sales from targeted research institutions to major clinical care organizations, focusing on patients with symptoms and diagnosed conditions, which dovetail with Nihon Kohden's marketing to doctors and cardiologists in Japan. The agreement comes on the heels of two strategic marketing deals concluded with Philips' local representative in Japan and with Medtronic Inc., both related to Itamar Medical's WatchPAT sleep diagnostic device.
"This agreement, along with the Philips agreement in the sleep breathing disorder market, will allow Itamar Medical to accelerate market penetration in Japan," said Itamar Medical CEO Gilad Glick. "With Japan constituting one of the major and most sophisticated medical device markets in the world, we consider Nihon Kohden to be an outstanding partner for accelerating clinical use of EndoPAT testing. The Japanese market is currently the most advanced in adapting clinical use of endothelial function assessment in the patient care strategy. Given the approved insurance reimbursement scheme for this test in Japan, the extensive ongoing academic and research activity there, and the inclusion of the EndoPAT test in the Japanese guidelines for assessing vascular function, we see significant potential in this deal."
Itamar Medical Ltd. is a publicly traded medical device company based in Caesarea’s Industrial Park, Israel. Since its founding in 1998, Itamar Medical has engaged in research and development of non-invasive medical devices for diagnosis of various medical conditions, including cardiovascular disease and sleep breathing disorders.
Nihon Kohden is manufactures, develops and distributes electronic medical devices in Japan. Nihon Kohden's international operations include branches in the United States, Europe and Asia, as well as distributors in other markets worldwide. In 2013, Nihon Kohden sales exceeded $1.4 billion. In Japan, Nihon Kohden's distribution network comprises of 120 offices throughout the country. Nihon Kohden is considered a provider of cardiology equipment (including ECG), emergency medical equipment, as well as sports healthcare and rehabilitation equipment.
The exclusive distribution agreement was reached after Nihon Kohden concluded that testing with Itamar Medical's EndoPAT device provides unique and important indications in assessing the risk of cardiovascular events. The agreement comes one year after Itamar Medical's subsidiary launched operations in Japan.
The EndoPAT device is used in non-invasive assessment of arterial health through the assessment of endothelial function (the inner lining of blood vessels), which regulates vasodilation and vasoconstriction. The test takes 15 minutes. A thimble-like sensor is placed on a patient finger on each hand and a blood pressure cuff temporarily stops blood flow to the arm to measure the arterial reaction and expansion once blood flow resumes. Unlike ultrasound-based tests, the EndoPAT device offers ease of use and accurate information that is not reliant on the competency of the testing staff and doctor who interprets the results.
"Nihon Kohden is the ideal partner for distribution of EndoPAT in Japan due to its stature among cardiologists and general practitioners," Itamar Medical vice president of Sales Eldad Singer noted. "The agreement, signed for three years, may be extended. It stipulates aggressive sales and growth targets as a condition for exclusivity."
Over the past year, Itamar Medical has diversified beyond sales from targeted research institutions to major clinical care organizations, focusing on patients with symptoms and diagnosed conditions, which dovetail with Nihon Kohden's marketing to doctors and cardiologists in Japan. The agreement comes on the heels of two strategic marketing deals concluded with Philips' local representative in Japan and with Medtronic Inc., both related to Itamar Medical's WatchPAT sleep diagnostic device.
"This agreement, along with the Philips agreement in the sleep breathing disorder market, will allow Itamar Medical to accelerate market penetration in Japan," said Itamar Medical CEO Gilad Glick. "With Japan constituting one of the major and most sophisticated medical device markets in the world, we consider Nihon Kohden to be an outstanding partner for accelerating clinical use of EndoPAT testing. The Japanese market is currently the most advanced in adapting clinical use of endothelial function assessment in the patient care strategy. Given the approved insurance reimbursement scheme for this test in Japan, the extensive ongoing academic and research activity there, and the inclusion of the EndoPAT test in the Japanese guidelines for assessing vascular function, we see significant potential in this deal."
Itamar Medical Ltd. is a publicly traded medical device company based in Caesarea’s Industrial Park, Israel. Since its founding in 1998, Itamar Medical has engaged in research and development of non-invasive medical devices for diagnosis of various medical conditions, including cardiovascular disease and sleep breathing disorders.
Nihon Kohden is manufactures, develops and distributes electronic medical devices in Japan. Nihon Kohden's international operations include branches in the United States, Europe and Asia, as well as distributors in other markets worldwide. In 2013, Nihon Kohden sales exceeded $1.4 billion. In Japan, Nihon Kohden's distribution network comprises of 120 offices throughout the country. Nihon Kohden is considered a provider of cardiology equipment (including ECG), emergency medical equipment, as well as sports healthcare and rehabilitation equipment.