09.11.14
Cordis Corp., a division of medical technology giant Johnson & Johnson, has launched its Incraft AAA stent graft system, an ultra-low profile device for use during endovascular aneurysm repair (EVAR) for patients suffering from infrarenal abdominal aortic aneurysms (AAA). The system is cleared for use and now available in Europe and Canada, Cordis officials reported recently.
According to the company, the device is an advancement in the EVAR field and provides a new option for physicians seeking a less-invasive treatment approach for AAA.
An estimated 24 million people worldwide suffer from AAA, an abnormal enlargement of the large blood vessel (aorta) that supplies blood to the abdomen, pelvis and legs. Left untreated, all aneurysms will eventually rupture and the majority of ruptured aneurysms result in death.
Most patients with AAA do not experience any noticeable symptoms, which is why AAA is commonly referred to as the “silent killer.” EVAR is a minimally invasive alternative to open surgery for the repair of an AAA. The procedure involves the placement of a stent graft into the aneurysm through a small incision in the groin to prevent the aneurysm from rupturing.
While the cause is not well-known, an aneurysm may develop in the lower part of the aorta and cause it to weaken as it enlarges or bulges. A damaged or ruptured AAA can cause life-threatening bleeding.
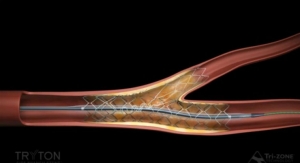
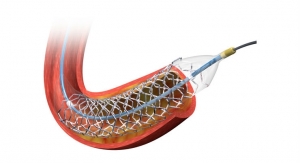
Incraft is intended for the endovascular treatment of patients with infrarenal AAA. The system is designed to reinforce the lower part of the aorta to prevent an aneurysm from rupturing. According to Cordis, Incraft is the lowest profile EVAR system now available in Europe and Canada with a 14 French (F) outer diameter, including the integrated sheath, which is equivalent to a 12 F catheter sheath introducer profile. Most EVAR stent grafts have a system profile ranging from 16 F to 22 F in size.
This ultra-low-profile device is designed for proximal and distal placement accuracy and allows for customization during the procedure to accommodate a wide range of anatomical sizes. This broad anatomical coverage is offered with a minimal number of product codes for easier pre-procedural planning.
“The Incraft system is an attractive new EVAR device option because its ultra-low profile design and customization allows physicians to consider this less invasive procedure for many patients, especially those with smaller vasculature who might otherwise be ineligible for EVAR,” said Giovanni Torsello, M.D., of the St. Franziskus Hospital Münster in Germany. “The recently published two-year data on the Incraft system from the Innovation trial in Europe demonstrated excellent performance adding to the scientific data supporting the device.”
The Innovation trial is a multi-center, open-label, prospective, non-randomized study designed to assess the safety and performance of the device in the treatment of patients with AAA with investigational sites in Germany and Italy. At two years, results from the study demonstrated the device performed well in patients and showed no incidences of aneurysm enlargement, endoleaks (type I or III), device or procedure related major adverse events, stent-graft migrations or stent fractures. One patient in the study developed a late graft occlusion unrelated to the device that was caused by shrinkage of the aneurysm. The two-year study results were presented at the 2014 Charing Cross Symposium and subsequently published in the July 2014 online issue of the Journal of Vascular Surgery.
“With the launch of the Incraft system, Cordis is bringing an innovative advancement to the field of EVAR, while entering a growth segment that further diversifies our strong product portfolio,” said Celine Martin, worldwide president, Cordis.
The system currently is approved for investigational device use only in the United States and Japan and is being studied in a global pivotal clinical study in the United States and Japan called the Inspiration trial, which completed enrollment in 2013.
Cordis develops interventional vascular technology.
According to a recent article in the Wall Street Journal, Cordis may not be part of the Johnson & Johnson family of companies for long. The beleaguered company, once the golden child among JNJ's portfolio of device companies, suffered during the slump of the drug-coated stent market in recent past. In 2011, JNJ officials said they would get out of the stent-making business.
The sales of Cordis could be worth $1.5 billion and $2 billion, according to unnamed sources cited by the Journal. The deal is expected to attract interest from private-equity firms and other healthcare companies.
According to the company, the device is an advancement in the EVAR field and provides a new option for physicians seeking a less-invasive treatment approach for AAA.
An estimated 24 million people worldwide suffer from AAA, an abnormal enlargement of the large blood vessel (aorta) that supplies blood to the abdomen, pelvis and legs. Left untreated, all aneurysms will eventually rupture and the majority of ruptured aneurysms result in death.
Most patients with AAA do not experience any noticeable symptoms, which is why AAA is commonly referred to as the “silent killer.” EVAR is a minimally invasive alternative to open surgery for the repair of an AAA. The procedure involves the placement of a stent graft into the aneurysm through a small incision in the groin to prevent the aneurysm from rupturing.
While the cause is not well-known, an aneurysm may develop in the lower part of the aorta and cause it to weaken as it enlarges or bulges. A damaged or ruptured AAA can cause life-threatening bleeding.
Incraft is intended for the endovascular treatment of patients with infrarenal AAA. The system is designed to reinforce the lower part of the aorta to prevent an aneurysm from rupturing. According to Cordis, Incraft is the lowest profile EVAR system now available in Europe and Canada with a 14 French (F) outer diameter, including the integrated sheath, which is equivalent to a 12 F catheter sheath introducer profile. Most EVAR stent grafts have a system profile ranging from 16 F to 22 F in size.
This ultra-low-profile device is designed for proximal and distal placement accuracy and allows for customization during the procedure to accommodate a wide range of anatomical sizes. This broad anatomical coverage is offered with a minimal number of product codes for easier pre-procedural planning.
“The Incraft system is an attractive new EVAR device option because its ultra-low profile design and customization allows physicians to consider this less invasive procedure for many patients, especially those with smaller vasculature who might otherwise be ineligible for EVAR,” said Giovanni Torsello, M.D., of the St. Franziskus Hospital Münster in Germany. “The recently published two-year data on the Incraft system from the Innovation trial in Europe demonstrated excellent performance adding to the scientific data supporting the device.”
The Innovation trial is a multi-center, open-label, prospective, non-randomized study designed to assess the safety and performance of the device in the treatment of patients with AAA with investigational sites in Germany and Italy. At two years, results from the study demonstrated the device performed well in patients and showed no incidences of aneurysm enlargement, endoleaks (type I or III), device or procedure related major adverse events, stent-graft migrations or stent fractures. One patient in the study developed a late graft occlusion unrelated to the device that was caused by shrinkage of the aneurysm. The two-year study results were presented at the 2014 Charing Cross Symposium and subsequently published in the July 2014 online issue of the Journal of Vascular Surgery.
“With the launch of the Incraft system, Cordis is bringing an innovative advancement to the field of EVAR, while entering a growth segment that further diversifies our strong product portfolio,” said Celine Martin, worldwide president, Cordis.
The system currently is approved for investigational device use only in the United States and Japan and is being studied in a global pivotal clinical study in the United States and Japan called the Inspiration trial, which completed enrollment in 2013.
Cordis develops interventional vascular technology.
According to a recent article in the Wall Street Journal, Cordis may not be part of the Johnson & Johnson family of companies for long. The beleaguered company, once the golden child among JNJ's portfolio of device companies, suffered during the slump of the drug-coated stent market in recent past. In 2011, JNJ officials said they would get out of the stent-making business.
The sales of Cordis could be worth $1.5 billion and $2 billion, according to unnamed sources cited by the Journal. The deal is expected to attract interest from private-equity firms and other healthcare companies.