09.05.14
Cothera LLC has received the CE mark for its portable VPULSE System.
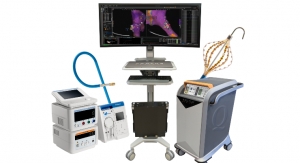
The VPULSE System combines intermittent pneumatic compression for deep vein thrombosis (DVT) prevention, dynamic compression therapy, and cold therapy into a single user, post-surgical rehabilitation device for hospital, clinic and home environments. The CE mark designation allows the VPULSE System to be sold in the European Union and other countries that recognize or require this approval.
“The VPULSE System combines DVT prevention and post orthopedic procedure rehabilitation and places these therapies and significant convenience in the hands of the patients,” Cothera General Manager Howard Edelman said. “This designation recognizes Cothera’s continued commitment to provide our customers with outstanding, high quality products and puts us on a path to increase international sales.”
DVT is among the most frequent hospital-acquired conditions. Though many professional associations and hospitals have developed protocols designed to prevent DVT and PE (pulmonary embolism), until now there has not been an economically feasible medical device that transitions from hospital to home use, thus allowing for continued patient compliance. Given the majority of DVT episodes occur post-discharge, the VPULSE System is available to help hospitals control readmission related costs, while supporting clinician treatment plans, the company claims.
Cothera also has received ISO 13485:2012 certification covering the design, development, production and of its medical devices, including the VPULSE System.
"As an early-stage medical device company, ISO 13485 certification represents a significant growth milestone," Edelman noted. "This designation, along with our CE Mark, broadens our path to increase international sales."
Founded in 2012, Cothera is a division of United Orthopedic Group Inc., based in Plano, Texas. The company was formed in response to market demands for better options to help prevent DVT and PE. The VPULSE System is sold internationally through medical device distributors.
The VPULSE System combines intermittent pneumatic compression for deep vein thrombosis (DVT) prevention, dynamic compression therapy, and cold therapy into a single user, post-surgical rehabilitation device for hospital, clinic and home environments. The CE mark designation allows the VPULSE System to be sold in the European Union and other countries that recognize or require this approval.
“The VPULSE System combines DVT prevention and post orthopedic procedure rehabilitation and places these therapies and significant convenience in the hands of the patients,” Cothera General Manager Howard Edelman said. “This designation recognizes Cothera’s continued commitment to provide our customers with outstanding, high quality products and puts us on a path to increase international sales.”
DVT is among the most frequent hospital-acquired conditions. Though many professional associations and hospitals have developed protocols designed to prevent DVT and PE (pulmonary embolism), until now there has not been an economically feasible medical device that transitions from hospital to home use, thus allowing for continued patient compliance. Given the majority of DVT episodes occur post-discharge, the VPULSE System is available to help hospitals control readmission related costs, while supporting clinician treatment plans, the company claims.
Cothera also has received ISO 13485:2012 certification covering the design, development, production and of its medical devices, including the VPULSE System.
"As an early-stage medical device company, ISO 13485 certification represents a significant growth milestone," Edelman noted. "This designation, along with our CE Mark, broadens our path to increase international sales."
Founded in 2012, Cothera is a division of United Orthopedic Group Inc., based in Plano, Texas. The company was formed in response to market demands for better options to help prevent DVT and PE. The VPULSE System is sold internationally through medical device distributors.