05.15.14
A clinical study sponsored by W.L. Gore & Associates Inc. has found the Gore Viabahn Endoprosthesis device with a Heparin bioactive surface is superior to percutaneous transluminal angioplasty (PTA), the current standard of care for treating stenoses or thrombotic occlusions. The company claims its Gore Revise Clinical study is the only randomized, controlled trial of stent grafts to investigate both stenotic and thrombotic occlusive arteriovenous (AV) access patients.
The multicenter, randomized-controlled trial evaluated the safety and effectiveness of the Gore Viabahn Endoprosthesis in treating stenoses or thrombotic occlusions of a synthetic AV access graft at the venous anastomosis. The study compared the results of two patient groups, one treated with the Gore Viabahn Device and the other receiving PTA. Key points from the Gore Revise clinical study:
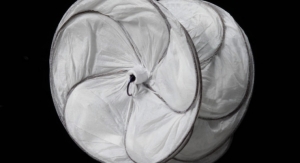
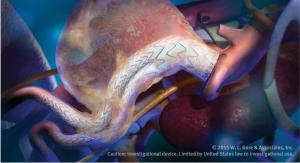
The Gore Viabahn Endoprosthesis is the lowest profile, most flexible, self-expanding, small-diameter, endoprosthesis available and the only stent or stent-graft to receive approval for the superficial femoral artery (SFA), iliac artery, and AV access, Gore contends. It is constructed with a durable, reinforced, biocompatible, expanded polytetrafluoroethylene (ePTFE) liner and attached to an external nitinol stent structure. The ePTFE luminal surface of the Gore Viabahn Endoprosthesis features the Carmeda BioActive Surface (CBAS Heparin Surface) for sustained anti-thrombotic bioactivity.
“With the Gore Revise Clinical Study, we wanted to set out and prove not only that the Gore Viabahn Endoprosthesis can successfully treat stenoses or thrombotic occlusions, but that the device is superior to the current standard of care,” said Ray Swinney, business unit leader for the Gore Peripheral Interventional Business Unit. “Investing in clinical studies such as Revise allows Gore to provide physicians with technology that is supported by trusted clinical data. In doing so, we can help physicians better serve the needs of their patients, particularly those with challenging anatomies that may not have been eligible for stent-graft procedures until now.”
The Gore Medical Products division has provided therapeutic solutions to complex medical problems for more than 35 years. During that time, more than 35 million innovative Gore Medical Devices have been implanted. The Gore Medical family of products includes vascular grafts, endovascular and interventional devices, surgical meshes for hernia repair, soft tissue reconstruction, staple line reinforcement and sutures for use in vascular, cardiac, and general surgery.The company is headquartered in Flagstaff, Ariz.
The multicenter, randomized-controlled trial evaluated the safety and effectiveness of the Gore Viabahn Endoprosthesis in treating stenoses or thrombotic occlusions of a synthetic AV access graft at the venous anastomosis. The study compared the results of two patient groups, one treated with the Gore Viabahn Device and the other receiving PTA. Key points from the Gore Revise clinical study:
- --- Statistical superiority over the PTA group in target lesion primary patency over a 24 month follow-up period (p = 0.008)
- --- Non-inferiority in terms of freedom from major device, treatment, or procedure related adverse events as compared to the PTA group (p < 0.001)
- --- Greater primary patency for both thrombotic and non-thrombotic patients as compared to PTA
- -- No reported fractures crossing the elbow
The Gore Viabahn Endoprosthesis is the lowest profile, most flexible, self-expanding, small-diameter, endoprosthesis available and the only stent or stent-graft to receive approval for the superficial femoral artery (SFA), iliac artery, and AV access, Gore contends. It is constructed with a durable, reinforced, biocompatible, expanded polytetrafluoroethylene (ePTFE) liner and attached to an external nitinol stent structure. The ePTFE luminal surface of the Gore Viabahn Endoprosthesis features the Carmeda BioActive Surface (CBAS Heparin Surface) for sustained anti-thrombotic bioactivity.
“With the Gore Revise Clinical Study, we wanted to set out and prove not only that the Gore Viabahn Endoprosthesis can successfully treat stenoses or thrombotic occlusions, but that the device is superior to the current standard of care,” said Ray Swinney, business unit leader for the Gore Peripheral Interventional Business Unit. “Investing in clinical studies such as Revise allows Gore to provide physicians with technology that is supported by trusted clinical data. In doing so, we can help physicians better serve the needs of their patients, particularly those with challenging anatomies that may not have been eligible for stent-graft procedures until now.”
The Gore Medical Products division has provided therapeutic solutions to complex medical problems for more than 35 years. During that time, more than 35 million innovative Gore Medical Devices have been implanted. The Gore Medical family of products includes vascular grafts, endovascular and interventional devices, surgical meshes for hernia repair, soft tissue reconstruction, staple line reinforcement and sutures for use in vascular, cardiac, and general surgery.The company is headquartered in Flagstaff, Ariz.