03.13.14
Marking a milestone in the evolution of fully-bioresorbable drug-eluting scaffolds for interventional cardiology, the first commercial implant of Elixir Medical Corporation’s CE Mark-approved DESolve Novolimus Eluting Coronary Scaffold was performed in Germany by Professor Dr. med. Holger Nef, head of the Cardiac Catheterization Laboratory, University Hospital in Giessen.
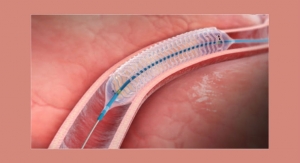
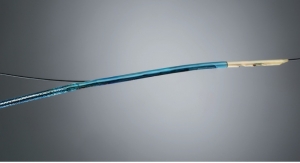
Elixir’s fully bioresorbable DESolve scaffold for coronary artery disease restores blood flow to the heart like metallic stents, but then dissolves to leave behind a treated artery that can function and move more naturally because it is free of a permanent implant. Developed from a proprietary poly-L Lactide (PLLA)-based polymer, DESolve provides optimal strength and support to the artery while delivering the novel anti-proliferative drug, Novolimus, Elixir claims.
The company contends the advantages of its DESolve scaffold system include: (a) maintaining radial strength and vessel support for the necessary period of vessel healing while degrading in one year; (b) increasing lumen area within six months; (c) self-apposing to the vessel wall up to the nominal deployed diameter; and (d) providing a wide margin of expansion.
“The fully bioresorbable DESolve System is easy to deliver. The acute performance of the scaffold demonstrated excellent stent apposition to the vessel wall and low recoil, and the patient is doing well,” said Professor Nef. Professor Dr. med. Christian Hamm, director of Cardiology at University Hospital Giessen. “I am confident that Elixir’s scaffold system will drive forward the field of vascular intervention.”
Developing an optimal bioresorbable scaffold had long been a challenge in the coronary vascular industry because it requires a level of strength and support that previously only permanent metallic stents had been able to provide while degrading in a timely manner and maintaining excellent clinical outcomes. The DESolve Novolimus Eluting Bioresorbable Coronary Scaffold overcomes these challenges and achieves vascular restoration within six months, reportedly years sooner than competitive scaffolds, Elixir said.
“Elixir’s DESolve has excellent clinical trial data to support its use. It is the first scaffold to degrade in one year, demonstrate lumen area increase within six months, and maintain the lumen area beyond one year,” said Stefan Verheye, M.D., Ph.D., ZNA Middleheim Hospital, Antwerp, Belgium, and co-principal investigator of the DESolve Nx trial. “These key differentiating features will create a paradigm shift in the treatment of patients with cardiovascular disease.”
The DESolve Nx pivotal trial enrolled 126 patients at 13 centers in Europe, Brazil and New Zealand. At one year, the DESolve Nx trial demonstrated a low MACE (Major Adverse Cardiac Events) rate of 5.69 percent with no definite scaffold thrombosis. Results using MSCT (Multi Slice Coherence Tomography), a noninvasive imaging modality to visualize coronary arteries and the manifestations of coronary artery disease, demonstrated that the lumen area measurements of treated vessels at one year were maintained to the results that were observed at six months using other imaging modalities.
“The fully bioresorbable DESolve Scaffold System holds the promise of transforming the interventional cardiology industry by raising the bar in clinical outcomes and leaving no permanent implant behind,” said Motasim Sirhan, president/CEO of Elixir Medical.
The fully bioresorbable DESolve Novolimus Eluting Scaffold System roll out will begin at initial centers in Europe. The scaffold is currently available in diameters of 3.0, 3.25 and 3.5 millimeters and lengths of 14, 18 and 28 millimeters. Additional sizes will be available this year.
Elixir Medical Corporation, a privately held company headquartered in Sunnyvale, Calif., develops drug-device combination products. The company's next-generation drug-eluting stent systems and bioresorbable coronary scaffold are designed to optimize localized drug delivery to provide safe and effective treatments for cardiovascular patients.
Elixir’s fully bioresorbable DESolve scaffold for coronary artery disease restores blood flow to the heart like metallic stents, but then dissolves to leave behind a treated artery that can function and move more naturally because it is free of a permanent implant. Developed from a proprietary poly-L Lactide (PLLA)-based polymer, DESolve provides optimal strength and support to the artery while delivering the novel anti-proliferative drug, Novolimus, Elixir claims.
The company contends the advantages of its DESolve scaffold system include: (a) maintaining radial strength and vessel support for the necessary period of vessel healing while degrading in one year; (b) increasing lumen area within six months; (c) self-apposing to the vessel wall up to the nominal deployed diameter; and (d) providing a wide margin of expansion.
“The fully bioresorbable DESolve System is easy to deliver. The acute performance of the scaffold demonstrated excellent stent apposition to the vessel wall and low recoil, and the patient is doing well,” said Professor Nef. Professor Dr. med. Christian Hamm, director of Cardiology at University Hospital Giessen. “I am confident that Elixir’s scaffold system will drive forward the field of vascular intervention.”
Developing an optimal bioresorbable scaffold had long been a challenge in the coronary vascular industry because it requires a level of strength and support that previously only permanent metallic stents had been able to provide while degrading in a timely manner and maintaining excellent clinical outcomes. The DESolve Novolimus Eluting Bioresorbable Coronary Scaffold overcomes these challenges and achieves vascular restoration within six months, reportedly years sooner than competitive scaffolds, Elixir said.
“Elixir’s DESolve has excellent clinical trial data to support its use. It is the first scaffold to degrade in one year, demonstrate lumen area increase within six months, and maintain the lumen area beyond one year,” said Stefan Verheye, M.D., Ph.D., ZNA Middleheim Hospital, Antwerp, Belgium, and co-principal investigator of the DESolve Nx trial. “These key differentiating features will create a paradigm shift in the treatment of patients with cardiovascular disease.”
The DESolve Nx pivotal trial enrolled 126 patients at 13 centers in Europe, Brazil and New Zealand. At one year, the DESolve Nx trial demonstrated a low MACE (Major Adverse Cardiac Events) rate of 5.69 percent with no definite scaffold thrombosis. Results using MSCT (Multi Slice Coherence Tomography), a noninvasive imaging modality to visualize coronary arteries and the manifestations of coronary artery disease, demonstrated that the lumen area measurements of treated vessels at one year were maintained to the results that were observed at six months using other imaging modalities.
“The fully bioresorbable DESolve Scaffold System holds the promise of transforming the interventional cardiology industry by raising the bar in clinical outcomes and leaving no permanent implant behind,” said Motasim Sirhan, president/CEO of Elixir Medical.
The fully bioresorbable DESolve Novolimus Eluting Scaffold System roll out will begin at initial centers in Europe. The scaffold is currently available in diameters of 3.0, 3.25 and 3.5 millimeters and lengths of 14, 18 and 28 millimeters. Additional sizes will be available this year.
Elixir Medical Corporation, a privately held company headquartered in Sunnyvale, Calif., develops drug-device combination products. The company's next-generation drug-eluting stent systems and bioresorbable coronary scaffold are designed to optimize localized drug delivery to provide safe and effective treatments for cardiovascular patients.