03.05.14
Medtronic Inc. continues to be haunted by its controversial bone graft product, Infuse.
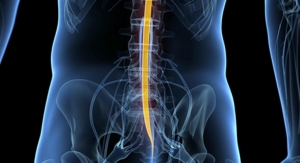
Nearly three years after the Minneapolis, Minn.-based company faced a mountain of criticism for overstating the bone morphogenetic protein's benefits and downplaying its risks, the controversial material is coming back to haunt Medtronic. The company admitted in a regulatory filing that it faces lawsuits from roughly 1,000 people over Infuse, also known as BMP-2 (human bone morphogenetic protein-2), a genetically engineered version of a protein the body produces naturally in small amounts to regulate bone growth and healing.
In mid-2011, a series of articles in The Spine Journal claimed that researchers with financial ties to Medtronic overstated the product’s benefits. One story accused researchers of slanting company-funded Infuse studies to favor its performance over a bone graft, the substance traditionally used in spinal fusions, and estimated the true incidence of adverse events to range from 10 to 50 percent, depending on the way in which the material is used. Side effects, according to the Spine Journal exposé, include male sterility, infection, bone loss and unwanted bone growth.
"When you start using a product 85 percent off label, there are going to be problems," Dan Spengler, a Vanderbilt University orthopedic surgeon, told the St.Paul (Minn.) Pioneer Press. He suspects many of the lawsuits involve patients who experienced excess bone growth that occurred after an off-label surgery.
Medtronic did not provide many details of the impending lawsuits in its regulatory filing, saying only that law firms have informed the company of the possibility of a large number of future Infuse claims.
Medtronic said, "Looking ahead, the company expects continued scientific and clinical research scrutiny focused on the safety and efficacy of Infuse in real-world, clinical experience."
At its height, Infuse generated sales of $800 million annually for Medtronic, but revenue has declined substantially since 2011.
Some court rulings have led to dismissals and others have limited claims, she said. "Medtronic stands behind Infuse bone graft and will vigorously defend it in court," she said.
In addition to the wave of lawsuits, several states are looking into sales and marketing practices involving Infuse.
Nearly three years after the Minneapolis, Minn.-based company faced a mountain of criticism for overstating the bone morphogenetic protein's benefits and downplaying its risks, the controversial material is coming back to haunt Medtronic. The company admitted in a regulatory filing that it faces lawsuits from roughly 1,000 people over Infuse, also known as BMP-2 (human bone morphogenetic protein-2), a genetically engineered version of a protein the body produces naturally in small amounts to regulate bone growth and healing.
In mid-2011, a series of articles in The Spine Journal claimed that researchers with financial ties to Medtronic overstated the product’s benefits. One story accused researchers of slanting company-funded Infuse studies to favor its performance over a bone graft, the substance traditionally used in spinal fusions, and estimated the true incidence of adverse events to range from 10 to 50 percent, depending on the way in which the material is used. Side effects, according to the Spine Journal exposé, include male sterility, infection, bone loss and unwanted bone growth.
Infuse has been implanted in the spines of more than 1 million people, most of whom received it in unapproved ways (off-label use). As much as 85 percent of BMP-2 use has been off-label, industry experts estimate.
"When you start using a product 85 percent off label, there are going to be problems," Dan Spengler, a Vanderbilt University orthopedic surgeon, told the St.Paul (Minn.) Pioneer Press. He suspects many of the lawsuits involve patients who experienced excess bone growth that occurred after an off-label surgery.
Medtronic did not provide many details of the impending lawsuits in its regulatory filing, saying only that law firms have informed the company of the possibility of a large number of future Infuse claims.
The company said it had not recorded any expense related to potential damages from the lawsuits.
"Additionally, the company cannot reasonably estimate the range of loss, if any, that may result from these matters," it said.
Medtronic said, "Looking ahead, the company expects continued scientific and clinical research scrutiny focused on the safety and efficacy of Infuse in real-world, clinical experience."
At its height, Infuse generated sales of $800 million annually for Medtronic, but revenue has declined substantially since 2011.
In a statement, company spokeswoman Cindy Resman said the cases are in early procedural stages and none had resulted in a finding of liability against Medtronic.
Some court rulings have led to dismissals and others have limited claims, she said. "Medtronic stands behind Infuse bone graft and will vigorously defend it in court," she said.
In addition to the wave of lawsuits, several states are looking into sales and marketing practices involving Infuse.
The patient lawsuits are the latest development in a decade-long saga that began shortly after the product debuted on the market in 2002, as doctors who received millions of dollars in royalties from Medtronic for other spine products began touting Infuse in medical journal articles they co-authored.
Eugene Carragee, head of spine surgery at Stanford University and editor of the Spine Journal, told thePioneer Press the lawsuits represent "a whole lot of people" who got a product that never really showed that it was more beneficial than methods used in traditional spinal fusion surgery.
In addition to the Spine Journal review, a damning investigation in 2012 by the U.S. Senate Committee on Finance of Infuse concluded that Medtronic executives secretly had drafted some of those favorable journal articles.
Over the course of 15 years, Medtronic paid $210 million to a group of 13 doctors and two corporations linked to doctors, including more than $34 million to University of Wisconsin orthopedic surgeon Thomas Zdeblick, who co-authored a series of papers about the product.
In 2013, two independent reports found that Infuse offered little benefit over conventional spine surgery and may be linked to serious harms including cancer and a complication that can cause sterility in men.
The product also can cause the growth of unwanted bone.