02.26.14
The Australian regulatory body for medical devices, the Therapeutic Goods Administration, has granted Minneapolis, Minn.-based IMRIS Inc. regulatory listing for Visius iCT, a ceiling mounted intraoperative computed tomography, allowing for sales and marketing in Australia.
“This is another step in our overall global launch following U.S. and European approvals,” said Jay D. Miller, IMRIS’ CEO and president. “Visius iCT offers the highest image quality of competing intraoperative systems along with market leading dose management capabilities. As procedures become less invasive, the need for better visualization with advanced imaging during procedures and low radiation exposure increases. With this unique solution, surgeons can confirm implant placements, fusion and other complex procedure results while still in the operating room.”
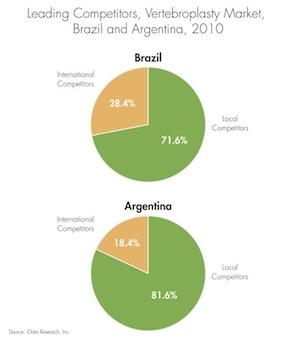
Market research shows that Australia is the second fastest growing spinal implant market behind China in the Asia Pacific region with a compounded annual growth rate of 8.4 percent since 2003. Australia is also the third largest contributor of medical imaging equipment sales in the area.
IMRIS currently has two Australian customers with Visius intraoperative MRI (iMRI) installations at Royal Children’s Hospital in Melbourne and Canberra Hospital in Canberra. In 2013, IMRIS established an office in Singapore to increase focus on the growing Asia Pacific market.
IMRIS received U.S. Food and Drug Administration (FDA) 510(k) and CE mark clearance for Visius iCT in July 2013. The company now has several systems sold to major U.S. neurosciences centers.
Visius iCT is designed to give personalized dose management together with diagnostic quality imaging during surgical procedures to assist surgeons in critical decision support and procedure guidance. The 64-slice scanner moves into and out of the operating room (OR) in a reported 30 seconds during surgery using ceiling-mounted rails to ease workflow. IMRIS also offers a configuration where the scanner can travel into two ORs to allow the hospital to use it for more patients.
Use of Visius iCT does not require patient transport or need for floor-mounted rails like other CT systems which opens up OR space and provides easier movement of surgical equipment. The surgical team can maintain familiar surgical protocols, workflow and infection control procedures.
In the interest of minimizing radiation exposure, Visius iCT features a suite of software applications such as 3-D volume rendering to aid in surgical planning and dose reduction algorithms which consider a patient’s unique characteristics and imaging target to maximize image quality and minimize the dose of radiation. The system software also offers state-of-the art dose tools including iterative reconstruction and real-time dose modulation.
IMRIS is focused on providing image guided therapy solutions through its Visius surgical theatre suite.
“This is another step in our overall global launch following U.S. and European approvals,” said Jay D. Miller, IMRIS’ CEO and president. “Visius iCT offers the highest image quality of competing intraoperative systems along with market leading dose management capabilities. As procedures become less invasive, the need for better visualization with advanced imaging during procedures and low radiation exposure increases. With this unique solution, surgeons can confirm implant placements, fusion and other complex procedure results while still in the operating room.”
Market research shows that Australia is the second fastest growing spinal implant market behind China in the Asia Pacific region with a compounded annual growth rate of 8.4 percent since 2003. Australia is also the third largest contributor of medical imaging equipment sales in the area.
IMRIS currently has two Australian customers with Visius intraoperative MRI (iMRI) installations at Royal Children’s Hospital in Melbourne and Canberra Hospital in Canberra. In 2013, IMRIS established an office in Singapore to increase focus on the growing Asia Pacific market.
IMRIS received U.S. Food and Drug Administration (FDA) 510(k) and CE mark clearance for Visius iCT in July 2013. The company now has several systems sold to major U.S. neurosciences centers.
Visius iCT is designed to give personalized dose management together with diagnostic quality imaging during surgical procedures to assist surgeons in critical decision support and procedure guidance. The 64-slice scanner moves into and out of the operating room (OR) in a reported 30 seconds during surgery using ceiling-mounted rails to ease workflow. IMRIS also offers a configuration where the scanner can travel into two ORs to allow the hospital to use it for more patients.
Use of Visius iCT does not require patient transport or need for floor-mounted rails like other CT systems which opens up OR space and provides easier movement of surgical equipment. The surgical team can maintain familiar surgical protocols, workflow and infection control procedures.
In the interest of minimizing radiation exposure, Visius iCT features a suite of software applications such as 3-D volume rendering to aid in surgical planning and dose reduction algorithms which consider a patient’s unique characteristics and imaging target to maximize image quality and minimize the dose of radiation. The system software also offers state-of-the art dose tools including iterative reconstruction and real-time dose modulation.
IMRIS is focused on providing image guided therapy solutions through its Visius surgical theatre suite.