02.06.14
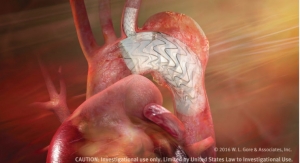
Flagstaff, Ariz.-based W. L. Gore & Associates Inc. is expanding the treatment range available in Australia and New Zealand for its Gore Excluder AAA endoprosthesis for used to treat abdominal aortic aneurysms (AAAs). The expanded offering includes lower profile components.
The 31 mm trunk-ipsilateral (on the same side of the body) component and 32 mm aortic extender will be used with an 18 Fr and 17 Fr Gore Dryseal sheath respectively, reduced from 20 Fr. For the contralateral (opposite side of body) legs, the reduced profile sizes allow the 12-20 mm contralateral leg component to be used with a 12 Fr Gore Dryseal sheath, the 23 mm to be used with a 14 Fr introducer sheath, and the 27 mm to be used with a 15 Fr introducer sheath. In addition, a new large diameter 35 mm trunk-ipsilateral leg and 36 mm aortic extender components will treat 30–32 mm vessel treatment range which expands overall treatment range to 19–32 mm. The 35 mm trunk-ipsilateral component and 36 mm aortic extender will be used with an 18 Fr Gore Dryseal sheath.
No changes have been made to the Gore Excluder device—instead, Gore has implemented an innovative process using ePTFE (polytetrafluoroethylene) materials to constrain the device onto the catheter. The lowering of the device profile exemplifies Gore’s commitment to improving patient safety while maintaining ease-of-use for the delivery of the Gore Excluder device.
“The availability of the expanded treatment range of the Gore Excluder device, in addition to the reduced profile components, provides physicians with a proven and durable option to better treat a broader range of patients diagnosed with abdominal aortic aneurysms,” said Stefan Ponosh, M.D., a vascular surgeon at Sir Charles Gairdner Hospital, Perth, Western Australia. “Reducing the profile of these devices allows more patients to benefit from minimally invasive access approaches to endovascular aortic repair.”
The Gore Excluder AAA endoprosthesis is an endovascular stent-graft designed to seal off the aneurysm and create a new path for blood flow. The device is inserted through a small incision in the patient’s leg using a catheter-based delivery technique. Once the physician has positioned the graft in the diseased aorta, the Gore C3 delivery system reportedly enables repositioning of the stent-graft. According to the company, the ability to reposition the device may minimize complications that could occur if the graft needs to be moved after the initial deployment.
The 31 mm trunk-ipsilateral (on the same side of the body) component and 32 mm aortic extender will be used with an 18 Fr and 17 Fr Gore Dryseal sheath respectively, reduced from 20 Fr. For the contralateral (opposite side of body) legs, the reduced profile sizes allow the 12-20 mm contralateral leg component to be used with a 12 Fr Gore Dryseal sheath, the 23 mm to be used with a 14 Fr introducer sheath, and the 27 mm to be used with a 15 Fr introducer sheath. In addition, a new large diameter 35 mm trunk-ipsilateral leg and 36 mm aortic extender components will treat 30–32 mm vessel treatment range which expands overall treatment range to 19–32 mm. The 35 mm trunk-ipsilateral component and 36 mm aortic extender will be used with an 18 Fr Gore Dryseal sheath.
No changes have been made to the Gore Excluder device—instead, Gore has implemented an innovative process using ePTFE (polytetrafluoroethylene) materials to constrain the device onto the catheter. The lowering of the device profile exemplifies Gore’s commitment to improving patient safety while maintaining ease-of-use for the delivery of the Gore Excluder device.
“The availability of the expanded treatment range of the Gore Excluder device, in addition to the reduced profile components, provides physicians with a proven and durable option to better treat a broader range of patients diagnosed with abdominal aortic aneurysms,” said Stefan Ponosh, M.D., a vascular surgeon at Sir Charles Gairdner Hospital, Perth, Western Australia. “Reducing the profile of these devices allows more patients to benefit from minimally invasive access approaches to endovascular aortic repair.”
The Gore Excluder AAA endoprosthesis is an endovascular stent-graft designed to seal off the aneurysm and create a new path for blood flow. The device is inserted through a small incision in the patient’s leg using a catheter-based delivery technique. Once the physician has positioned the graft in the diseased aorta, the Gore C3 delivery system reportedly enables repositioning of the stent-graft. According to the company, the ability to reposition the device may minimize complications that could occur if the graft needs to be moved after the initial deployment.