01.27.14
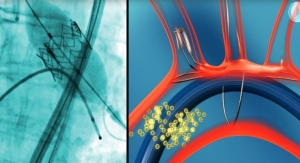
Bolton Medical, a medical device manufacturer which specializes in endovascular treatments for aortic repair, has initiated its Phase II U.S. clinical trial to study the safety and effectiveness of the Treovance Abdominal Stent-Graft with Navitel Delivery System. The first two cases were performed late last fall by John Henretta, M.D., and Mike Douglas, M.D., vascular surgeons at Carolina Vascular, Mission Hospital in Asheville, N.C.
"Both cases went really well. It's a device that allows for precise placement. Its delivery system is easy to advance through the iliac anatomy, and the material is non-porous. I feel secure about the device's long term-durability and we look forward to enrolling more patients," Henretta said.
A third case was performed around the same time as the first two by Andres Schanzer, M.D., a vascular surgeon at the University of Massachusetts Medical Center in Worcester, Mass.
"The first Phase II Treovance endovascular aneurysm repairs mark an exciting time for Bolton Medical and the investigators involved in this trial. We look forward to many more implants and the opportunity to treat patients," said Matthew Eagleton, M.D., a vascular surgeon with the Department of Vascular Surgery within the Sydell and Arnold Miller Family Heart & Vascular Institute at Cleveland Clinic and national principal investigator for the Phase II U.S. Treovance AAA Stent-Graft Clinical Trial.
The primary objective of the clinical study is to assess the safety and effectiveness of the Treovance Abdominal Stent-Graft in subjects with infrarenal aortic aneurysms. Results of this study will support approval of the product in the United States. Phase II of the study will include 150 patients enrolled at 30 institutions located throughout the United States.
Features of the Treovance Abdominal Stent-Graft include a three-piece, adaptable system that can accommodate a wide-range of anatomies; active fixation through suprarenal and infrarenal barbs permitting proximal and supplemental fixation in highly angulated necks; and a highly flexible design, conformable even in difficult anatomies.
The Treovance Abdominal Stent-Graft is offered with the Navitel Delivery System, an intuitive low profile system featuring mechanical advantage and a completely detachable sheath assembly. The Navitel Delivery System also includes the proximal clasping system of Bolton Medical's Relay Thoracic Stent-Graft, allowing for accurate deployment.
Bolton Medical currently manufactures Relay and Relay NBS Thoracic Stent-Grafts. They are indicated for the treatment of thoracic aortic pathologies such as aneurysms, penetrating ulcers, pseudoaneurysms, and intramural hematomas in adult patients. To date, more than 10,000 Relay and Relay NBS Stent-Grafts have been implanted worldwide, according to the company. Relay was awarded U.S. Food and Drug Administration approval on Sept. 21, 2012.
Bolton Medical is a privately held company, with international operations based in Barcelona, Spain, and U.S. operations in Sunrise, Fla. Bolton Medical is a wholly-owned subsidiary of the Werfen Life Group, a diverse manufacturer and supplier of products for the medical industry.
"Both cases went really well. It's a device that allows for precise placement. Its delivery system is easy to advance through the iliac anatomy, and the material is non-porous. I feel secure about the device's long term-durability and we look forward to enrolling more patients," Henretta said.
A third case was performed around the same time as the first two by Andres Schanzer, M.D., a vascular surgeon at the University of Massachusetts Medical Center in Worcester, Mass.
"The first Phase II Treovance endovascular aneurysm repairs mark an exciting time for Bolton Medical and the investigators involved in this trial. We look forward to many more implants and the opportunity to treat patients," said Matthew Eagleton, M.D., a vascular surgeon with the Department of Vascular Surgery within the Sydell and Arnold Miller Family Heart & Vascular Institute at Cleveland Clinic and national principal investigator for the Phase II U.S. Treovance AAA Stent-Graft Clinical Trial.
The primary objective of the clinical study is to assess the safety and effectiveness of the Treovance Abdominal Stent-Graft in subjects with infrarenal aortic aneurysms. Results of this study will support approval of the product in the United States. Phase II of the study will include 150 patients enrolled at 30 institutions located throughout the United States.
Features of the Treovance Abdominal Stent-Graft include a three-piece, adaptable system that can accommodate a wide-range of anatomies; active fixation through suprarenal and infrarenal barbs permitting proximal and supplemental fixation in highly angulated necks; and a highly flexible design, conformable even in difficult anatomies.
The Treovance Abdominal Stent-Graft is offered with the Navitel Delivery System, an intuitive low profile system featuring mechanical advantage and a completely detachable sheath assembly. The Navitel Delivery System also includes the proximal clasping system of Bolton Medical's Relay Thoracic Stent-Graft, allowing for accurate deployment.
Bolton Medical currently manufactures Relay and Relay NBS Thoracic Stent-Grafts. They are indicated for the treatment of thoracic aortic pathologies such as aneurysms, penetrating ulcers, pseudoaneurysms, and intramural hematomas in adult patients. To date, more than 10,000 Relay and Relay NBS Stent-Grafts have been implanted worldwide, according to the company. Relay was awarded U.S. Food and Drug Administration approval on Sept. 21, 2012.
Bolton Medical is a privately held company, with international operations based in Barcelona, Spain, and U.S. operations in Sunrise, Fla. Bolton Medical is a wholly-owned subsidiary of the Werfen Life Group, a diverse manufacturer and supplier of products for the medical industry.