01.10.14
Veniti Medical has been granted CE Mark approval for its Vici Venous stent, used to treat occluded iliofemoral veins, as well as the Veniti Veni RF Plus Ablation System, for varicose veins.
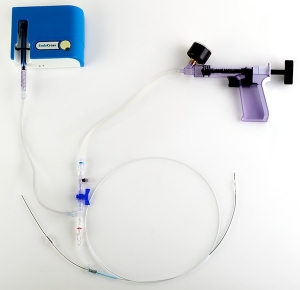
The Vici Venous Stent is the first stent designed and intended from its inception as a dedicated venous stent. "We believe the unique combination of end-to-end crush resistance, flexibility and continuous vein wall coverage make the Vici Venous Stent particularly well-suited for treating iliofemoral venous obstruction," said Scott Solano, Veniti President/CEO. "After many years of using re-purposed arterial stents to treat this serious disease that affects so many people, we are excited to offer physicians a stent designed specifically for iliofemoral venous obstruction. We are eager to demonstrate the Vici Venous Stent advantages to physicians and to provide them with a venous-specific stent."
Coupled with a bidirectional delivery system, the Vici will give physicians better accuracy and more options in terms of placement, the company claims.
The Veni RF Plus system can be used to treat the approximately 20 million Americans suffering from venous insufficiency. It works by vaporizing saline solution to ablate the veins, eliminating the need for compression of the vein. Veniti also anticipates other varicosities will be eliminated as the saline vapor travels beyond the application point.
Veniti Medical will continue studies for the Vici venous stent and will apply for U.S. Food and Drug Administration (FDA) approval upon completion of clinical trials.
Veniti is a provider of medical devices that treat venous disease. The Veniti Vidi Vena Cava Filter is in early stages of clinical studies in New Zealand, and the company is seeking approval from the FDA to begin clinical studies of the Vidi Vena Cava Filter in the United States. Veniti's research and development and manufacturing is conducted in Fremont, Calif., while the company's headquarters is in St. Louis, Mo.
The Vici Venous Stent is the first stent designed and intended from its inception as a dedicated venous stent. "We believe the unique combination of end-to-end crush resistance, flexibility and continuous vein wall coverage make the Vici Venous Stent particularly well-suited for treating iliofemoral venous obstruction," said Scott Solano, Veniti President/CEO. "After many years of using re-purposed arterial stents to treat this serious disease that affects so many people, we are excited to offer physicians a stent designed specifically for iliofemoral venous obstruction. We are eager to demonstrate the Vici Venous Stent advantages to physicians and to provide them with a venous-specific stent."
Coupled with a bidirectional delivery system, the Vici will give physicians better accuracy and more options in terms of placement, the company claims.
The Veni RF Plus system can be used to treat the approximately 20 million Americans suffering from venous insufficiency. It works by vaporizing saline solution to ablate the veins, eliminating the need for compression of the vein. Veniti also anticipates other varicosities will be eliminated as the saline vapor travels beyond the application point.
Veniti Medical will continue studies for the Vici venous stent and will apply for U.S. Food and Drug Administration (FDA) approval upon completion of clinical trials.
Veniti is a provider of medical devices that treat venous disease. The Veniti Vidi Vena Cava Filter is in early stages of clinical studies in New Zealand, and the company is seeking approval from the FDA to begin clinical studies of the Vidi Vena Cava Filter in the United States. Veniti's research and development and manufacturing is conducted in Fremont, Calif., while the company's headquarters is in St. Louis, Mo.