12.11.13
Mobius Imaging LLC, based in Ayer, Mass., has received ISO 13485 certification for its quality management system.
"Achieving ISO certification was a priority since the company's inception. Our goal was not only to comply with all FDA (U.S. Food and Drug Administration) guidance and regulations, but also to go a step beyond. We view this accomplishment as significant progress toward meeting the demand for Mobius Imaging products and recognize the importance of the ISO standard in marketing Mobius products internationally," said CEO Gene Gregerson.
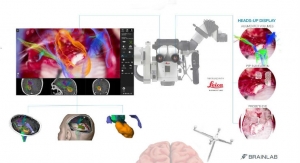
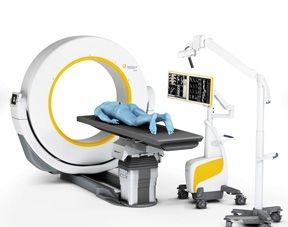
ISO 13485 recognizes a company’s quality management system for the design and manufacture of medical devices. It demonstrates a commitment from management for quality throughout the product development lifecycle and the product's lifespan with customers, company officials noted. The certification also is “an important milestone” toward the commercialization of the company's first product, the Airo Mobile computed tomography (CT) imaging system, Gregerson noted. The Airo Mobile CT system is distributed under an exclusive sales and service agreement with Brainlab.
Mobius uses what it calls “intelligent imaging” to develop novel products that are engineered specifically to intuitively acclimate into different care environments to further enable quality care.
"Our decision to become ISO certified was fueled by our passion for providing our customers with the very best products and services in the medical imaging industry," said Gregerson. "Mobius' certification to this standard further validates that our products are manufactured at the highest level of quality control."
"Achieving ISO certification was a priority since the company's inception. Our goal was not only to comply with all FDA (U.S. Food and Drug Administration) guidance and regulations, but also to go a step beyond. We view this accomplishment as significant progress toward meeting the demand for Mobius Imaging products and recognize the importance of the ISO standard in marketing Mobius products internationally," said CEO Gene Gregerson.
ISO 13485 recognizes a company’s quality management system for the design and manufacture of medical devices. It demonstrates a commitment from management for quality throughout the product development lifecycle and the product's lifespan with customers, company officials noted. The certification also is “an important milestone” toward the commercialization of the company's first product, the Airo Mobile computed tomography (CT) imaging system, Gregerson noted. The Airo Mobile CT system is distributed under an exclusive sales and service agreement with Brainlab.
Mobius uses what it calls “intelligent imaging” to develop novel products that are engineered specifically to intuitively acclimate into different care environments to further enable quality care.
"Our decision to become ISO certified was fueled by our passion for providing our customers with the very best products and services in the medical imaging industry," said Gregerson. "Mobius' certification to this standard further validates that our products are manufactured at the highest level of quality control."