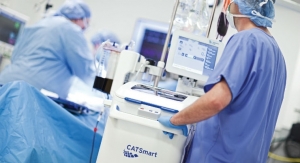
The blood-focused division of the Japanese med tech giant has signed a deal with the U.S. Department of Defense to share costs in developing a device that can treat donated blood used in emergency transfusions.
Under the agreement, the Defense Department will spend up to $14.8 million to help Terumo get its Mirasol System through the U.S. Food and Drug Administration process, guaranteeing $3.5 million over the next three years with the option of contributing another $11.3 million to fund safety and efficacy studies. In turn, Terumo will contribute up to $15.1 million to the project.
Together, the two plan to develop a device that uses riboflavin and ultraviolet light to treat whole blood and render disease-causing viruses less pathogenic, eventually shipping Mirasol out to deployed military forces for use in combat transfusions.
Terumo executives said the partnership allows the company to cost-effectively develop a life-saving tool whose benefits could stretch far beyond the battlefield.
"The Mirasol system for whole blood provides the potential for safe blood products in environments where this is not possible or practical today," Raymond Goodrich, Ph.D., vice president of Scientific and Clinical Affairs for Terumo BCT, said in a statement. "Its use could change the way in which blood is made available to people on a global basis."
Mirasol has been closely aligned with the U.S. government since its inception, and the latest agreement follows a $3.5 million grant awarded in 2012 to bankroll early trials of the device and about $10.6 million in federal grant and appropriations funding from 2009. If the Department of Defense exercises its full funding option, Mirasol's development will tap about $30 million in government cash.
"Enhancing the safety of military blood products while retaining therapeutic efficacy is critical to the treatment of wounded service members on the battlefield, and is among the highest priorities of the Defense Medical Research and Development Program," said Major Charles DiTusa, product manager, U.S. Army Medical Material Development Activity. "Under this contract, Terumo BCT will perform critical studies toward FDA clearance of the Mirasol system for whole blood, a pathogen reduction device intended to decrease the risk of blood transfusion associated with bacteria, viruses, parasites and white blood cells."