11.15.13
Edwards Lifesciences Corporation has expanded its heart valve manufacturing facility in Changi North Crescent, Singapore, to accommodate a reported growing worldwide demand for tissue heart valves.
“We warmly welcome Edwards’ decision to expand its facility here, commented Yeoh Keat Chuan, managing director of the Singapore Economic Development Board. “This is a strong affirmation of Singapore’s competitiveness as a location for advanced medical technology manufacturing.”
This expansion of Edwards’ Singapore manufacturing facility more than doubles the size of the current building to approximately 24,000 square meters, while adding offices and laboratories, a large clean room and training facilities. With this new project, Edwards has invested approximately S$95 million in Singapore since 2005.
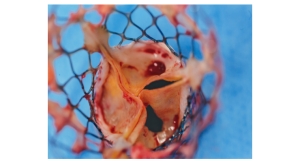
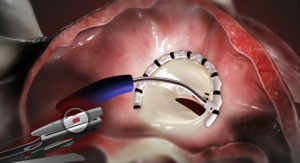
As part of Edwards’ global heart valve manufacturing program, the state-of-the-art Singapore facility contributes to the production of Edwards’ surgical and transcatheter tissue heart valves. The company also operates facilities dedicated to manufacturing heart valves at its global headquarters in Irvine, Calif. and in Horw, Switzerland.
“Edwards’ Singapore facility was developed to meet the global demands for our technologically advanced tissue heart valve replacement products and to help us serve the needs of patients worldwide,” said Donald E. Bobo, Jr., Edwards’ corporate vice president, heart valve therapy. “We are proud that our continued investment in Singapore will result in employment opportunities for hundreds of additional workers to join the 1,000 employees currently working for Edwards in the region. We are very grateful to the Economic Development Board for their extraordinary efforts in working with us to ensure this success.”
Edwards Lifesciences makes devices for the heart valves and hemodynamic monitoring segments.
“We warmly welcome Edwards’ decision to expand its facility here, commented Yeoh Keat Chuan, managing director of the Singapore Economic Development Board. “This is a strong affirmation of Singapore’s competitiveness as a location for advanced medical technology manufacturing.”
This expansion of Edwards’ Singapore manufacturing facility more than doubles the size of the current building to approximately 24,000 square meters, while adding offices and laboratories, a large clean room and training facilities. With this new project, Edwards has invested approximately S$95 million in Singapore since 2005.
As part of Edwards’ global heart valve manufacturing program, the state-of-the-art Singapore facility contributes to the production of Edwards’ surgical and transcatheter tissue heart valves. The company also operates facilities dedicated to manufacturing heart valves at its global headquarters in Irvine, Calif. and in Horw, Switzerland.
“Edwards’ Singapore facility was developed to meet the global demands for our technologically advanced tissue heart valve replacement products and to help us serve the needs of patients worldwide,” said Donald E. Bobo, Jr., Edwards’ corporate vice president, heart valve therapy. “We are proud that our continued investment in Singapore will result in employment opportunities for hundreds of additional workers to join the 1,000 employees currently working for Edwards in the region. We are very grateful to the Economic Development Board for their extraordinary efforts in working with us to ensure this success.”
Edwards Lifesciences makes devices for the heart valves and hemodynamic monitoring segments.