11.08.13
Biotronik SE & Co. KG's DREAMS are about to come true.
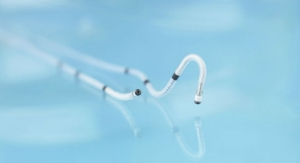
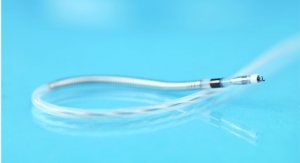
The German manufacturer of cardio- and endovascular medical technology has implanted its DRug Eluting Absorbable Metal Scaffold (DREAMS) in its first patient as part of the clinical study Biosolve II. Prof. Dr. Michael Haude from the Lukaskrankenhaus, Neuss, Germany, conducted the procedure.
DREAMS is a bioabsorbable scaffold that combines the mechanical advantages of a metallic stent with a reliable bioabsorption profile that opens vessels and keeps them from re-clogging while avoiding the long-term disadvantages of permanent metal stents, which cage the vessel segment over the course of the patient's life. The DREAMS scaffold, by contrast, is designed to be absorbed over time, leaving a naturally restored vessel. The scaffold is made of a bioabsorbable magnesium alloy and coated with a bioabsorbable polymer matrix and an anti-proliferative Limus drug.
Building on the results of the Biosolve I study, which gave Biotronik valuable insight into the clinical workings of the first generation of DREAMS, Biosolve II is a prospective, multi-center clinical trial evaluating the safety and efficacy of DREAMS in its improved design. Approximately 120 patients are expected to be enrolled, with follow-up investigations performed at one, six, 12, 24, and 36 months. The primary endpoint is in-segment late lumen loss (LLL). This study will be used for regulatory approval of the device.
"I was impressed by the ease of deliverability and vessel adaptability," Prof. Haude said. "Even in quite a challenging lesion like this with significant calcification, the acute performance was similar to a contemporary drug-eluting stent, but offering the advantage of uncaging the vessel in the long run."
"As a global pioneer in bioabsorbable magnesium, Biotronik has refined this technology based on physicians' input to deliver the optimal balance between vessel scaffolding, absorption profile, and drug elution characteristics," noted Dr. Daniel Buehler, President of Biotronik's Vascular Intervention division. "With the clinical data of our first generation in the Biosolve I study, and the preclinical data of this new generation, I am confident that this treatment will prove its efficacy."
The Biosolve II study will enroll patients in Germany, Belgium, Denmark, The Netherlands, Switzerland, Spain, Brazil, and Singapore.
Biotronik is headquartered in Berlin, Germany, and has a global workforce of more than 5,600 employees. Its U.S. headquarters is located in Lake Oswego, Ore.
The German manufacturer of cardio- and endovascular medical technology has implanted its DRug Eluting Absorbable Metal Scaffold (DREAMS) in its first patient as part of the clinical study Biosolve II. Prof. Dr. Michael Haude from the Lukaskrankenhaus, Neuss, Germany, conducted the procedure.
DREAMS is a bioabsorbable scaffold that combines the mechanical advantages of a metallic stent with a reliable bioabsorption profile that opens vessels and keeps them from re-clogging while avoiding the long-term disadvantages of permanent metal stents, which cage the vessel segment over the course of the patient's life. The DREAMS scaffold, by contrast, is designed to be absorbed over time, leaving a naturally restored vessel. The scaffold is made of a bioabsorbable magnesium alloy and coated with a bioabsorbable polymer matrix and an anti-proliferative Limus drug.
Building on the results of the Biosolve I study, which gave Biotronik valuable insight into the clinical workings of the first generation of DREAMS, Biosolve II is a prospective, multi-center clinical trial evaluating the safety and efficacy of DREAMS in its improved design. Approximately 120 patients are expected to be enrolled, with follow-up investigations performed at one, six, 12, 24, and 36 months. The primary endpoint is in-segment late lumen loss (LLL). This study will be used for regulatory approval of the device.
"I was impressed by the ease of deliverability and vessel adaptability," Prof. Haude said. "Even in quite a challenging lesion like this with significant calcification, the acute performance was similar to a contemporary drug-eluting stent, but offering the advantage of uncaging the vessel in the long run."
"As a global pioneer in bioabsorbable magnesium, Biotronik has refined this technology based on physicians' input to deliver the optimal balance between vessel scaffolding, absorption profile, and drug elution characteristics," noted Dr. Daniel Buehler, President of Biotronik's Vascular Intervention division. "With the clinical data of our first generation in the Biosolve I study, and the preclinical data of this new generation, I am confident that this treatment will prove its efficacy."
The Biosolve II study will enroll patients in Germany, Belgium, Denmark, The Netherlands, Switzerland, Spain, Brazil, and Singapore.
Biotronik is headquartered in Berlin, Germany, and has a global workforce of more than 5,600 employees. Its U.S. headquarters is located in Lake Oswego, Ore.