10.01.13
The Defense Advanced Research Projects Agency (DARPA) has exercised an option agreement to proceed with the third year of a five-year $6.8 million contract that was awarded to San Diego, Calif.-based Aethlon Medical Inc. on Sept. 30, 2011, under DARPA's Dialysis-Like Therapeutics (DLT) program.
Aethlon develops selective therapeutic filtration devices to address infectious disease, cancer and other life-threatening conditions.
The third year of Aethlon's DLT contract contains eight milestones representing a potential of $1,534,099 in revenue opportunity, the company reported. To date, Aethlon has invoiced $3,188,131 to DARPA for achieving fourteen of sixteen milestone objectives targeted in the first two years of the DLT program.
The goal of the DLT program is to develop a portable device that removes "dirty" blood from the body, separates harmful agents, and returns "clean" blood to the body in a manner similar to dialysis treatment of kidney failure. The resulting device would decrease the morbidity and mortality of sepsis, thereby saving thousands of lives and billions of dollars in the United States annually.
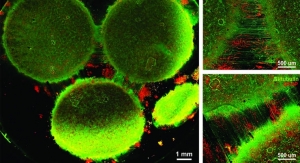
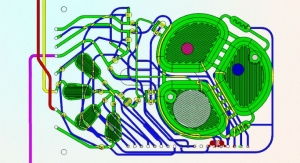
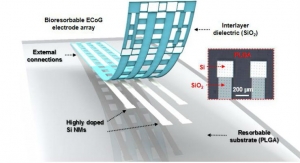
In the DLT program, Aethlon has been contracted to use its ADAPT system to create an extracorporeal blood purification cartridge that selectively eliminates sepsis-enabling particles from circulation to promote recovery and prevent sepsis. ADAPT converges advanced plasma membrane technology with high-affinity drug agents to allow the selective, yet rapid clearance of disease targets from the entire circulatory system without damaging blood cells or removing particles essential for health, according to the company. Aethlon also has been contracted to develop a novel blood circulatory instrument that will deliver ADAPT based and other therapeutic filtration devices without systemic anticoagulants normally required in extracorporeal therapies.
Beyond the civilian need for anti-sepsis therapies, the device proposed in the DLT program would play an important role in saving the lives of wounded U.S. military personnel, as infection leading to sepsis is a significant cause of mortality. When sepsis is complicated by shock, approximately half of patients do not survive for 30 days, even if effective antibiotics are used. Unfortunately, the fatality rate from sepsis can be high, given that antibiotic-resistant bacteria are an increasing problem for injured military personnel and military treatment facilities. Bio-agents engineered for resistance against antibiotics also represent a significant threat to both military troops and citizens. Current culture-based methods of identifying blood-borne pathogens can take 48 hours or longer to identify the offending pathogen, and some blood-borne pathogens do not propagate in culture. Pending these culture results, septic patients are treated with protocol-based broad-spectrum antibiotics. In the event the offending pathogen is resistant to the empirically chosen antibiotic, the fatality rate may increase as much as 9 percent per hour.
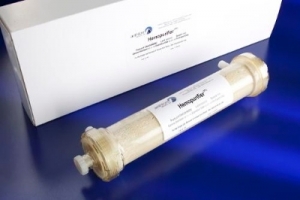
Aethlon currently is preparing to launch the first U.S. studies of its Hemopurifier therapy based on the recent approval of an investigational device exemption (IDE) by the U.S. Food and Drug Administration. Under the IDE, Aethlon will enroll 10 end-stage renal disease patients infected with Hepatitis C (HCV) as part of a feasibility study to demonstrate Hemopurifier safety. Successful completion of the feasibility study will set the stage for Aethlon to conduct pivotal studies required for market clearance to treat HCV and other disease conditions.
Aethlon develops selective therapeutic filtration devices to address infectious disease, cancer and other life-threatening conditions.
The third year of Aethlon's DLT contract contains eight milestones representing a potential of $1,534,099 in revenue opportunity, the company reported. To date, Aethlon has invoiced $3,188,131 to DARPA for achieving fourteen of sixteen milestone objectives targeted in the first two years of the DLT program.
The goal of the DLT program is to develop a portable device that removes "dirty" blood from the body, separates harmful agents, and returns "clean" blood to the body in a manner similar to dialysis treatment of kidney failure. The resulting device would decrease the morbidity and mortality of sepsis, thereby saving thousands of lives and billions of dollars in the United States annually.
In the DLT program, Aethlon has been contracted to use its ADAPT system to create an extracorporeal blood purification cartridge that selectively eliminates sepsis-enabling particles from circulation to promote recovery and prevent sepsis. ADAPT converges advanced plasma membrane technology with high-affinity drug agents to allow the selective, yet rapid clearance of disease targets from the entire circulatory system without damaging blood cells or removing particles essential for health, according to the company. Aethlon also has been contracted to develop a novel blood circulatory instrument that will deliver ADAPT based and other therapeutic filtration devices without systemic anticoagulants normally required in extracorporeal therapies.
Beyond the civilian need for anti-sepsis therapies, the device proposed in the DLT program would play an important role in saving the lives of wounded U.S. military personnel, as infection leading to sepsis is a significant cause of mortality. When sepsis is complicated by shock, approximately half of patients do not survive for 30 days, even if effective antibiotics are used. Unfortunately, the fatality rate from sepsis can be high, given that antibiotic-resistant bacteria are an increasing problem for injured military personnel and military treatment facilities. Bio-agents engineered for resistance against antibiotics also represent a significant threat to both military troops and citizens. Current culture-based methods of identifying blood-borne pathogens can take 48 hours or longer to identify the offending pathogen, and some blood-borne pathogens do not propagate in culture. Pending these culture results, septic patients are treated with protocol-based broad-spectrum antibiotics. In the event the offending pathogen is resistant to the empirically chosen antibiotic, the fatality rate may increase as much as 9 percent per hour.
Aethlon currently is preparing to launch the first U.S. studies of its Hemopurifier therapy based on the recent approval of an investigational device exemption (IDE) by the U.S. Food and Drug Administration. Under the IDE, Aethlon will enroll 10 end-stage renal disease patients infected with Hepatitis C (HCV) as part of a feasibility study to demonstrate Hemopurifier safety. Successful completion of the feasibility study will set the stage for Aethlon to conduct pivotal studies required for market clearance to treat HCV and other disease conditions.