08.21.13
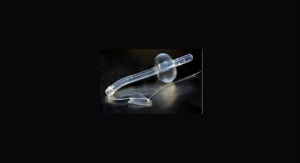
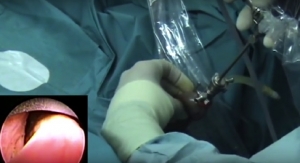
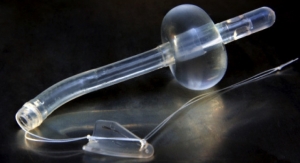
Maple Grove, Minn.-based NxThera Inc. has closed on a round of Series C financing which brought in $18.2 million. The company also was awarded the CE mark for its Rezum system to treat benign prostatic hyperplasia (BPH). Company officials said they will use this new influx of capital to conduct a U.S. clinical trial in pursuance of U.S. Food and Drug Administration clearance for Rezum; begin targeted, early commercialization of its Rezum system in selected European countries in 2014; and expand its applied research and feasibility studies to evaluate applications of its sterile water vapor technology to treat prostate cancer and kidney cancer.
“We sincerely appreciate the continued engagement and support of our investors as we move closer to realizing NxThera’s vision of a fundamentally new treatment approach that will transform the experience of urologists and the millions of men they treat who suffer from prostate conditions,” said Bob Paulson, president and CEO of NxThera. “With this round of financing, we are well-positioned to undertake and complete our U.S. BPH pivotal trial, obtain early commercial BPH experience in Europe, and accelerate our prostate and kidney cancer clinical development programs.”
In aging men, the normal flow of urine from the bladder often is restricted by bladder obstructions, often as the result of enlarged prostate tissue or other urethral strictures. Symptoms of BPH include increased urination frequency, sudden urges to urinate, and weak urine flow. According to data gathered by an independent research organization hired by NxThera, more than 12 million men in the United States have been diagnosed with BPH and more than 70 percent of men over age 60 experience symptoms of the disease. More than 2.5 million men in the United States have been diagnosed with prostate cancer.
NxThera is focused on applying its sterile water vapor technology platform in a variety of endourology conditions including BPH, prostate cancer and kidney cancer.
“We sincerely appreciate the continued engagement and support of our investors as we move closer to realizing NxThera’s vision of a fundamentally new treatment approach that will transform the experience of urologists and the millions of men they treat who suffer from prostate conditions,” said Bob Paulson, president and CEO of NxThera. “With this round of financing, we are well-positioned to undertake and complete our U.S. BPH pivotal trial, obtain early commercial BPH experience in Europe, and accelerate our prostate and kidney cancer clinical development programs.”
In aging men, the normal flow of urine from the bladder often is restricted by bladder obstructions, often as the result of enlarged prostate tissue or other urethral strictures. Symptoms of BPH include increased urination frequency, sudden urges to urinate, and weak urine flow. According to data gathered by an independent research organization hired by NxThera, more than 12 million men in the United States have been diagnosed with BPH and more than 70 percent of men over age 60 experience symptoms of the disease. More than 2.5 million men in the United States have been diagnosed with prostate cancer.
NxThera is focused on applying its sterile water vapor technology platform in a variety of endourology conditions including BPH, prostate cancer and kidney cancer.