Niki Arrowsmith05.09.13
The U.S. Food and Drug Administration granted 510(k) clearance to Masimo for its new Rainbow Acoustic Monitoring sensor, the RAS 125-c acoustic respiration cloth sensor. Previously, the device was cleared for adult use, but the new clearance is for the continuous, noninvasive monitoring of acoustic respiration rate in pediatric patients.
Respiration rate is a critical vital sign that provides early detection of respiratory compromise and patient distress. Continuous monitoring of respiration rate is especially important for post-surgical patients receiving patient-controlled analgesia for pain management as the sedation can induce respiratory depression and place patients at considerable risk of serious injury or death. Last year The Joint Commission issued a “Sentinel Event Alert” on opioid-induced respiratory depression and recommended continuous monitoring of oxygenation and ventilation of post-surgical patients—a sentinel event, as defined by the Commission, is an unexpected occurrence involving death or serious physical or psychological injury, or the risk thereof. Similarly, Anaesthesia Patient Safety Foundation guidelines include oxygenation and ventilation monitoring in all patients receiving opioids. According to a 2007 Anesthesiology journal article, traditional methods for respiration rate monitoring—nasal cannula capnography—historically have been somewhat unreliable and difficult for some patients to tolerate.
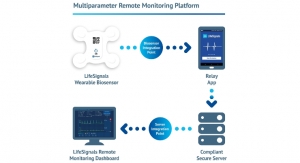
Masimo Rainbow Acoustic Monitoring is designed to noninvasively and continuously measures respiration rate using an adhesive sensor with an integrated acoustic transducer that is applied to the patient’s neck, according to the company. Now indicated for use in pediatric patients, the accuracy of the RAS-125c Acoustic Respiration Rate Sensor remains the same (as the adult version) at plus or minus one over the range of four to 70 breaths per minute.
“It’s going to be a home run,” said Michael Ramsay, M.D., chief of the department of anesthesiology and pain management at Baylor University Medical Center in Dallas, Texas, said of acoustic respiration rate. “Kids don’t tolerate capnography,” he added. Capnography entails a nasal cannula—a tube inserted into the nasal passage—for monitoring respiratory rate and end-tidal carbon dioxide. Ramsay recently finished a study on the efficacy of acoustic respiration rate that will soon be published in Anesthesia & Analgesia.
“We have been eager to offer this patient-friendly respiration rate monitoring solution to pediatric patients in the United States,” said Masimo founder and CEO Joe Kiani. “We believe that noninvasive, continuous monitoring of acoustic respiration rate is better suited for younger patients than traditional capnography technologies that we also offer. And because the RAS-125c acoustic respiration rate sensor is more comfortable and tolerable for patients, it will help clinicians improve patient outcomes.”
Headquartered in Irvine, Calif., Masimo makes non-invasive products for patient monitoring.
Respiration rate is a critical vital sign that provides early detection of respiratory compromise and patient distress. Continuous monitoring of respiration rate is especially important for post-surgical patients receiving patient-controlled analgesia for pain management as the sedation can induce respiratory depression and place patients at considerable risk of serious injury or death. Last year The Joint Commission issued a “Sentinel Event Alert” on opioid-induced respiratory depression and recommended continuous monitoring of oxygenation and ventilation of post-surgical patients—a sentinel event, as defined by the Commission, is an unexpected occurrence involving death or serious physical or psychological injury, or the risk thereof. Similarly, Anaesthesia Patient Safety Foundation guidelines include oxygenation and ventilation monitoring in all patients receiving opioids. According to a 2007 Anesthesiology journal article, traditional methods for respiration rate monitoring—nasal cannula capnography—historically have been somewhat unreliable and difficult for some patients to tolerate.
Masimo Rainbow Acoustic Monitoring is designed to noninvasively and continuously measures respiration rate using an adhesive sensor with an integrated acoustic transducer that is applied to the patient’s neck, according to the company. Now indicated for use in pediatric patients, the accuracy of the RAS-125c Acoustic Respiration Rate Sensor remains the same (as the adult version) at plus or minus one over the range of four to 70 breaths per minute.
“It’s going to be a home run,” said Michael Ramsay, M.D., chief of the department of anesthesiology and pain management at Baylor University Medical Center in Dallas, Texas, said of acoustic respiration rate. “Kids don’t tolerate capnography,” he added. Capnography entails a nasal cannula—a tube inserted into the nasal passage—for monitoring respiratory rate and end-tidal carbon dioxide. Ramsay recently finished a study on the efficacy of acoustic respiration rate that will soon be published in Anesthesia & Analgesia.
“We have been eager to offer this patient-friendly respiration rate monitoring solution to pediatric patients in the United States,” said Masimo founder and CEO Joe Kiani. “We believe that noninvasive, continuous monitoring of acoustic respiration rate is better suited for younger patients than traditional capnography technologies that we also offer. And because the RAS-125c acoustic respiration rate sensor is more comfortable and tolerable for patients, it will help clinicians improve patient outcomes.”
Headquartered in Irvine, Calif., Masimo makes non-invasive products for patient monitoring.