Hardy Diagnostics Releases Rapid Test for COVID-19
By Newswire | 03.25.20
Partnership with Autobio Diagnostics enables company to become a U.S. supplier of one-step lateral flow assay that detects both IgG and IgM antibodies.
UPDATE: The manufacturer has reached out to MPO so we would let our readers know this specific product has been discontinued and is no longer available.
--------------------
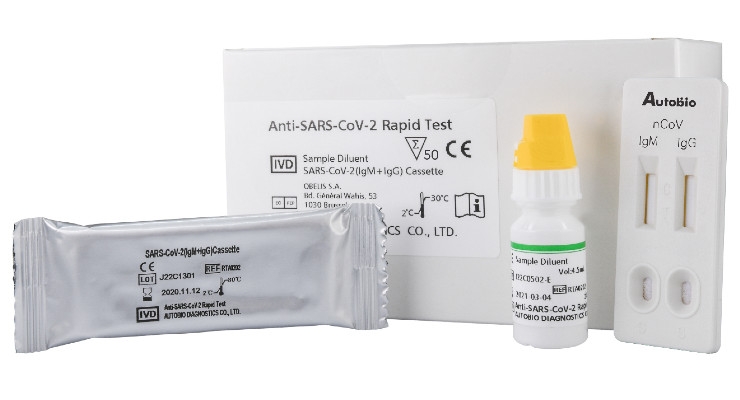
Hardy Diagnostics, a medical device manufacturer based in Santa Maria, California, announced it has begun accepting orders for a new in vitro diagnostic medical device: Anti-SARS-CoV-2 Rapid Test (Cat. No. RTA0203).
This immunoassay is intended specifically for determining the possibility of a COVID-19 infection and was developed by Autobio Diagnostics Co. Ltd., a publicly traded major microbiology medical device manufacturer, based in Zhengzhou, China. Through this partnership, Hardy Diagnostics and Autobio have begun to open supply chains to deliver this rapid test to the United States. This rapid market deployment of a new in vitro diagnostic medical device was made possible through the FDA Emergency Use Authorization (EUA) program. This program is used in times of crisis, such as the current COVID-19 pandemic. It allows for simultaneous commercialization while the Anti-SARS-CoV-2 Rapid Test is under review, which enables Hardy Diagnostics to supply product during this critical time.
Anti-SARS-CoV-2 Rapid Test is a rapid, one-step lateral flow assay intended for the presumptive qualitative detection of IgM and IgG antibodies to the SARS-CoV-2 virus in patients suspected of a COVID-19 infection.
IgM antibodies are generated initially by the body as a result of infection at about the time symptoms appear. They will dissipate within approximately one month. IgG antibodies are generated by the body about one week after symptoms appear and last for an extended amount of time.
By using a patient’s finger-prick blood, serum or plasma specimen, the Anti-SARS-CoV-2 Rapid Test offers a turnaround time of only 15 minutes. This simple-to-use test requires no equipment or special expertise or training to implement.
“We are incredibly proud of the work our partners in China have accomplished,” said Andre Hsiung, director of technical services at Hardy Diagnostics. “Because Autobio quickly developed this technology and because the FDA allowed Emergency Use Authorization, we will be able to more effectively leverage our sales network to get this product out to where it is needed the most.”
Anti-SARS-CoV-2 Rapid Test is an immunoassay intended for the presumptive qualitative detection of IgM and IgG antibodies to the SARS-CoV-2 virus in plasma from anticoagulated human blood (Heparin / EDTA / sodium citrate), serum or capillary finger-prick blood (no anticoagulants or additives) from individuals with signs and symptoms of infection who are suspected of COVID-19 infection. Testing is limited to certified laboratories able to perform highly complex tests at point-of-care or in the laboratory.
The Anti-SARS-CoV-2 Rapid Test is intended for use by clinical laboratory personnel specifically instructed and trained in the techniques of in vitro diagnostic procedures.
Negative results do not rule out SARS-CoV-2 infection, particularly in those who have been in recent contact with the virus, due to the lag time between exposure and the patient’s antibody response. Positive results may be due to past or present infections with SARS-CoV-2 coronavirus strains, such as coronavirus HKU1, NL63, OC43 or 229E.
This test should not be used as the sole basis for patient management decisions. Test results must be combined with clinical observations, patient history, and epidemiological information.
Follow-up testing with a molecular diagnostic test should be considered to confirm the infection status. This test is not intended for the screening of donated blood.
This test will be useful in identifying asymptomatic and mildly symptomatic carriers of the SARS-CoV-2 virus. It will also be helpful in identifying persons who were infected by the virus previously, but may not have been properly diagnosed.
Serological assays are of critical importance to determine sero-prevalence in a given population, to define previous exposure, and to identify highly reactive human donors for the generation of convalescent serum as therapeutics. Sensitive and specific identification of coronavirus SARS-CoV-2 antibody titers will also support screening of health care workers to identify those who are already immune and can be deployed to care for infected patients, thus minimizing the risk of viral spread to colleagues and other patients.
--------------------
Hardy Diagnostics, a medical device manufacturer based in Santa Maria, California, announced it has begun accepting orders for a new in vitro diagnostic medical device: Anti-SARS-CoV-2 Rapid Test (Cat. No. RTA0203).
This immunoassay is intended specifically for determining the possibility of a COVID-19 infection and was developed by Autobio Diagnostics Co. Ltd., a publicly traded major microbiology medical device manufacturer, based in Zhengzhou, China. Through this partnership, Hardy Diagnostics and Autobio have begun to open supply chains to deliver this rapid test to the United States. This rapid market deployment of a new in vitro diagnostic medical device was made possible through the FDA Emergency Use Authorization (EUA) program. This program is used in times of crisis, such as the current COVID-19 pandemic. It allows for simultaneous commercialization while the Anti-SARS-CoV-2 Rapid Test is under review, which enables Hardy Diagnostics to supply product during this critical time.
Anti-SARS-CoV-2 Rapid Test is a rapid, one-step lateral flow assay intended for the presumptive qualitative detection of IgM and IgG antibodies to the SARS-CoV-2 virus in patients suspected of a COVID-19 infection.
IgM antibodies are generated initially by the body as a result of infection at about the time symptoms appear. They will dissipate within approximately one month. IgG antibodies are generated by the body about one week after symptoms appear and last for an extended amount of time.
By using a patient’s finger-prick blood, serum or plasma specimen, the Anti-SARS-CoV-2 Rapid Test offers a turnaround time of only 15 minutes. This simple-to-use test requires no equipment or special expertise or training to implement.
“We are incredibly proud of the work our partners in China have accomplished,” said Andre Hsiung, director of technical services at Hardy Diagnostics. “Because Autobio quickly developed this technology and because the FDA allowed Emergency Use Authorization, we will be able to more effectively leverage our sales network to get this product out to where it is needed the most.”
Anti-SARS-CoV-2 Rapid Test is an immunoassay intended for the presumptive qualitative detection of IgM and IgG antibodies to the SARS-CoV-2 virus in plasma from anticoagulated human blood (Heparin / EDTA / sodium citrate), serum or capillary finger-prick blood (no anticoagulants or additives) from individuals with signs and symptoms of infection who are suspected of COVID-19 infection. Testing is limited to certified laboratories able to perform highly complex tests at point-of-care or in the laboratory.
The Anti-SARS-CoV-2 Rapid Test is intended for use by clinical laboratory personnel specifically instructed and trained in the techniques of in vitro diagnostic procedures.
Negative results do not rule out SARS-CoV-2 infection, particularly in those who have been in recent contact with the virus, due to the lag time between exposure and the patient’s antibody response. Positive results may be due to past or present infections with SARS-CoV-2 coronavirus strains, such as coronavirus HKU1, NL63, OC43 or 229E.
This test should not be used as the sole basis for patient management decisions. Test results must be combined with clinical observations, patient history, and epidemiological information.
Follow-up testing with a molecular diagnostic test should be considered to confirm the infection status. This test is not intended for the screening of donated blood.
This test will be useful in identifying asymptomatic and mildly symptomatic carriers of the SARS-CoV-2 virus. It will also be helpful in identifying persons who were infected by the virus previously, but may not have been properly diagnosed.
Serological assays are of critical importance to determine sero-prevalence in a given population, to define previous exposure, and to identify highly reactive human donors for the generation of convalescent serum as therapeutics. Sensitive and specific identification of coronavirus SARS-CoV-2 antibody titers will also support screening of health care workers to identify those who are already immune and can be deployed to care for infected patients, thus minimizing the risk of viral spread to colleagues and other patients.