YOU SEARCHED BY...
SEARCH BY...
Country
- China(2)
- Singapore(1)
- Switzerland(2)
- United Kingdom(1)
- United States(42)
Outsourcing Directory
Company Headquarters
501 County Road E2 Extension
New Brighton, MN 55112
United States
Company Description
Donatelle offers medical component and device manufacturing through product development, prototyping, tool design and fabrication, injection molding, micromolding, micro-MIM, bioresorbable molding, liquid silicone molding, precision machining, device assembly, packaging and sterilization management.
Outsourcing Directory
- Assembly Services
- Automation
- Bar Coding
- CNC Milling
- Component Parts
- Contract Mfg
- Design/Engineering
- Electrical Discharge Machining (EDM)
- Failure Analysis
- FDA Compliance
- Full-Service Manufacturing
- GMP Compliance
- Grinding Machines
- Hot Stamping
- Implant Manufacturing
- Labeling
- Laser Marking
- Machining, Finishing Metals
- Machining, Finishing Plastics
- Micro-abrasive Blasting
- Mold Building
- Molding, Implantables
- Molding, Metal Injection (MIM)
- Molding, Micro
- Molding, Plastics
- Molding, Silicone
- Molding, Thermoplastic Elastomers
- Packaging, Bagging/Pouching
- Packaging, Cartoning
- Packaging, Consulting/Design
- Precision Machining
- Product Development
- Project Management
- Rapid Prototyping
- Research and Development
- Stamping
- Statistical Analysis
- Strategic Planning
- Supply Chain Management
- Tooling
- Welding
- Welding, Ultrasonic
In-Vitro Diagnostic Categories
Medical Device Categories
Content From Donatelle
-
Data Driven Process Development in Medical Device Manufacturing—A Medtech Makers Q&A
Data can be a valuable tool to help ensure accuracy in manufacturing of components for medical devices.Sponsored Content Released on 09.19.2022
-
Successfully Incorporating Remote Audits Post-Pandemic—A Medtech Makers Q&A
The pandemic caused remote audits to become the norm for a period of time, but lessons learned could keep the practice in place.Sponsored Content Released on 05.23.2022
-
Addressing Manufacturing Challenges with Automation—A Medtech Makers Q&A
A combination of factors are causing medical device manufacturers to reevaluate the benefits of a supply partner with automated solutions in place.Sponsored Content Released on 02.28.2022
-
Miniaturizing Metal Through Injection Molding—A Medtech Makers Q&A
Metal injection molding for micro components offers numerous benefits, but the right supplier is critical.Sponsored Content Released on 10.11.2021
-
Developing Next Generation Implants with Bioresorbables—A Medtech Makers Q&A
To help restore natural anatomy following a medical procedure, some device makers are curious about bioresorbable molded devices.Sponsored Content Released on 08.16.2021
-
Expediting Time to Market—A Medtech Makers Q&A
Donatelle’s Mike Kaiser and Todd Owens provide insights on speeding up development to get products out to market more quickly.Sponsored Content Released on 09.30.2020
-
Micro-MIM Succeeds At Making Precise Micro Metal Parts
...White Papers / Tech Papers Released on 09.16.2020
-
How To Conquer The Challenges Of Micro-Manufacturing
By Raghu VadlamudiPeople In The News Released on 08.11.2020
-
The Criticality of Process Validation
By: Todd Owens and Raghu VadlamudiPeople In The News Released on 08.06.2020
-
How To Avoid Compliance & Timeline Risks When Selecting A Medical Device Supplier
By: Matt KnutsonWhite Papers / Tech Papers Released on 06.25.2020
Related Content
-
Materials | Molding
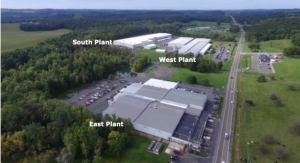
Tessy Plastics Expands
The expansion is planned to be part of the “South Plant” located in Elbridge, New York.Tessy Plastics 01.06.20
-
Tubing & Extrusion

-
Materials

Understanding Key Processes for Mixing APIs with Silicone
The pros and cons of drug delivery and device enhancement.Andrew Gaillard, Healthcare & Medical Segment Manager, Trelleborg Sealing Solutions 12.05.19
-
Tubing & Extrusion
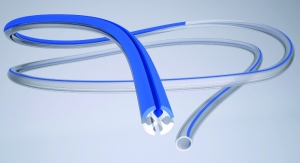
5 Questions for Trelleborg Healthcare & Medical at Medica/CompaMed 2019
Engineering and material expertise including geometric transition extrusion technology.Sean Fenske, Editor-in-Chief 11.15.19
-
3D/Additive Manufacturing | Machining & Laser Processing | Molding
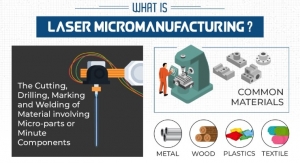
Laser Micromanufacturing 101—Everything You Need to Know
Laser micromanufacturing is used for a variety of purposes including additive manufacturing, CNC machining, and injection molding.Laser Light Technologies
-

Toray Provides a Cost-Efficient Materials Option for Liquid-Contact Medical Device Manufacturers
The company's TOYOLAC resin has robust chemical resistance and is sterilizable but cannot be autoclaved.Toray Industries Inc. 11.06.19
-

Flambeau Hires VP of North American Operations
New executive previously worked as vice president of Global Manufacturing for Terumo BCT.Flambeau Inc. 11.05.19
-













