YOU SEARCHED BY...
SEARCH BY...
Outsourcing Directory
Country
- Canada(2)
- China(2)
- Costa Rica(1)
- France(2)
- Germany(2)
- India(3)
- Italia (2)
- Japan(1)
- Lithuania(1)
- Mexico(1)
- New Zealand(1)
- Singapore(3)
- Switzerland(3)
- Taiwan(4)
- The Dominican Republic(1)
- United Kingdom(3)
- United States(342)
Outsourcing Directory
American Kuhne, a brand of Graham Engineering Corp.
Company Headquarters
1203 Eden Road
York, PA 17402
United States
Outsourcing Directory
In-Vitro Diagnostic Categories
Medical Device Categories
Related Content
-
Diagnostics | Digital Health | Neurological

6 Proposals Receive Seed Funds to Develop Medical Devices for Children
Projects address cystic fibrosis, urinary flow disorders, kidney reflux, brain injury, skeletal defects & vision impairment.Childrens Hospital of Philadelphia 07.31.18
-

-
Contract Manufacturing | Digital Health | Electronics | R&D & Design

Electronics Integration into Disposable and Single-Use Medical Devices
A number of critical factors need to be considered in order to ensure a successful implementation.Rick Walker, GM Product Design and Development, & Meredith Canty, Director Drug Delivery Systems, at SMC Ltd. 06.29.18
-
Digital Health

What a Long, Strange Trip It’s Been…
...Maria Shepherd, President and Founder, Medi-Vantage 06.04.18
-

Cardiac Dimensions Names New VP of Clinical and Regulatory Affairs
Also, M.H. Carnegie & Co.’s Trevor Moody joins Cardiac Dimensions’ Board of Directors.Business Wire 05.08.18
-
India’s New Guidelines Will Ultimately Benefit Local Players
Move follows the price caps set in 2017 on coronary stent and knee replacement implant products.GlobalData 05.07.18
-
R&D & Design

To Clinical or Not to Clinical, That Is the Question
...David C. Robson, Principal, Robson Advisors 05.03.18
-

Three Conspirators Sentenced to Prison in $16.6 Million Fraudulent Medical Device Scheme
The trio marketed laser devices to elderly consumers as a cure-all.U.S. Department of Justice 04.27.18
-
Tubing & Extrusion
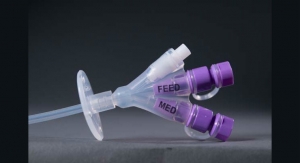
Mastering Matings: Reengineering the Enteral Feeding Luer Connector
A new ISO standard for medical tubing connectors inspires one company to take on a leading role in development.Caroline Lee, Account Representative, Mintz & Hoke 04.11.18
-

India’s New Medical Devices Rules
The country’s MDR has eight provisions every manufacturer should know if interested in doing business in India.Blake E. Wilson and Shilpa Prem, Hogan Lovells 04.03.18
-
Contract Manufacturing | Materials | Molding
Deliberate Disposal: Single-Use & Disposables Technologies
Single-use or disposable devices can help reduce infections and save costs for healthcare facilities and manufacturers.Sam Brusco, Associate Editor 04.03.18
-
Software & IT

Accruent Acquires Connectiv, Broadens Healthcare Technology Management Portfolio
Asset management solution addresses medical device cyber risks.Business Wire 03.21.18
-
Materials

Practical Matters: A Look at Medical Device Materials
Changing healthcare regulations and hospitals’ war on germs are driving innovation in medical device materials.Michael Barbella, Managing Editor 03.15.18
-
Materials

Material World: Is Titanium the New PEEK?
...Dawn A. Lissy, President & Founder, Empirical 03.14.18
-

Aziyo, Cook Biotech Further Expand the Bio Envelope Franchise Into Additional Markets
New agreements provide for use of Bio Envelope in neuromodulation markets.Business Wire 03.13.18











