Sam Brusco, Associate Editor11.07.17
For a very long time, the surgical biopsy—a sample of tissue taken from the body for examination—has been the diagnostic standard. Biopsies are most commonly done to look for cancer after tissue abnormalities are discovered—for example, if an ultrasound or MRI reveals a lump or mass, or a mole on the skin has changed shape. Nearly all of these involve using a sharp tool to remove the tissue in question. They can be CT-guided to minimize discomfort, but are always invasive to some degree.
As a result of medicine’s push toward minimally invasive procedures, an alternative, far less painful biopsy method has emerged. Liquid biopsy enables doctors to discover a wealth of information about a tumor through a blood sample (i.e., liquid), rather than a surgical procedure. Fragments of DNA deposited into the bloodstream by tumors have shown success in non-invasive screening for early-stage cancers, monitoring cancer treatment response, and explaining why some cancers resist therapies.
“Procedures of the traditional tissue biopsy are painful, associated with complications such as potential infections, and often available only in central hospitals which require patients to travel long distances,” former MIT postdoc Mohammad Qasaimeh, who was involved in a study in which a microfluidic technique was used to capture and count circulating plasma cells from small samples of blood to diagnose and track multiple myeloma progression, said in a press release. “Capturing plasma cells from blood samples can serve as a liquid biopsy, which can be performed in clinics as often as required, and serve as a diagnostic and prognostic test during and after chemotherapy treatment. Moreover, captured cells can be used for drug testing and thus serve as a tool for personalized medicine.”
This in vitro diagnostic (IVD) method has the potential to transform cancer care, but it’s still relatively early in development. Taking and testing tissue samples still remains the standard, and a number of questions would need to be addressed in clinical trials before making liquid biopsy common practice: How much does test accuracy vary among tumor types and cancer stages? Is the sampling representative of all the genetic clones in a tumor, or is there bias toward particular sub-regions? Until these are answered, liquid biopsy will remain in the clinical research stage.
Established IVD technologies and devices are implemented in either clinical or over-the-counter environments to examine biological samples like blood, saliva, urine, or tissue collected from the body. These devices include chemicals and reagents, analytical equipment, disposables (cartridges or test strips), and software supporting the analytical processes in the lab.
“In the IVD industry, there is growing interest in increasing the portfolio of complete clinical solutions,” illuminated Mario Koksch M.D., Ph.D., MBA, vice president and general manager, Flow Cytometry Business Unit of Indianapolis, Ind.-based Beckman Coulter Life Sciences, part of Danaher Corporation. Dr. Koksch is responsible for the global flow cytometry business, strategy, and manufacturing centers in Marseille, France; Bangalore, India; and Suzhou, China. “A complete clinical solution includes reagents, controls, calibrators, an automated sample prep, and automated data analysis approach. These complete solutions allow for higher quality results in a broader range of labs in less time, which makes initial diagnostics of disease states, monitoring the disease’s progress, and selecting the most appropriate treatment options for individual patients more straightforward, rational, and objective.”
The IVD market is currently undergoing a transformation. Large instruments in central labs are seeing migration to tabletop and hand-held devices, particularly in areas like next-generation sequencing, molecular diagnostics, and point-of-care (PoC). IVD testing has been migrating from the hospital to the physician’s office, and in some cases, to the home. This trend has resulted in a degree of industry consolidation, evidenced by a few significant mergers and acquisitions over the past year. There is also strong pressure to control costs because reimbursement can be challenging for IVD technologies. Because of this and other factors, IVD labs must be incredibly efficient to keep costs down.
“We see a number of increasing challenges facing IVD laboratories,” commented Christine Snyder, senior director of strategic marketing for Diagnostic Systems at Becton, Dickinson and Company (BD), a Franklin Lakes, N.J.-based firm that develops, manufactures, and sells medical supplies, devices, laboratory equipment, and diagnostic products worldwide. “The changing reimbursement landscape, shortage of qualified laboratory technologists, and ongoing consolidation of laboratories means larger organizations are serving more customers with fewer employees. With that, our customers continue to focus on providing fast and accurate test results with greater efficiency, which means we are seeing the widespread adoption of laboratory automation and informatics platforms, products and services. Increasing use of informatics technology helps our customers better manage laboratory information flow and can improve communication between laboratories and physicians.”
Instant Interpretation
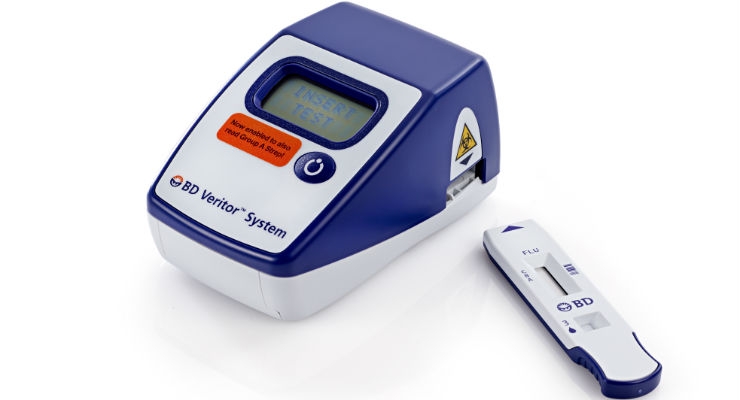
The modern world craves immediate results. The medical extension of this need is realized in the impetus for technology to diagnose diseases at the PoC, rather than using traditional lab-based tests. PoC IVD devices fit the bill for this fast-paced attitude because they are usually portable, relatively simple to operate, and the instruments return results rapidly.
Lab-based tests require an order to draw blood, followed by the patient’s trip to the draw site. The specimen is then sent for analysis to the lab, after which the result is finally communicated to the clinician, then finally to the patient or caregiver. Despite labs’ attempts to minimize this turnaround time, it can sometimes take a day or more to receive results. PoC diagnostic systems shortcut the steps of collecting and shipping a specimen; test results can be achieved on the spot in the doctor’s office or at home.
“In the past, in vitro diagnostics were only available in the clinical setting,” recalled Ted Rowan, regional sales manager of Boyd Technologies, a Lee, Mass.-based international supplier and precision converter of advanced flexible materials for the medical device and life sciences markets. “Advances in technology have made it possible to take many tests at home, often with nearly instant results. Home tests are no less accurate, more convenient, and easily performed by the end user. PoC systems are generally membrane-based test strips enclosed inside a plastic cassette. These patient self-tests can detect changes in disease pathophysiology at the earliest onset, empowering patients to take better control of their illnesses.”
“Existing and startup companies manufacturing PoC instruments and kits are advancing and growing rapidly,” observed Craig Occhiato, microfluidics team manager for Bürkert Fluid Control Systems, a Charlotte, N.C.-based manufacturer of measurement and control systems for liquids and gases for the medical device industry. “This is changing the convenience, time, and cost of the IVD landscape—moving it from the large lab and hospital lab to a doctor’s office, patient room, emergency medicine setting, or even a third world country.”
PoC diagnostic technologies generally fall into two categories. The first are small handheld devices with accompanying test strips or cartridges that offer qualitative or quantitative analysis, for example a glucose biosensor fingerstick or a small cartridge device for blood gas analysis. Larger bench-top PoC devices are essentially smaller, less complex versions of laboratory instruments, for example critical care analyzers or small hematology and immunology analyzers.
“When ITL first began work in the field over 40 years ago, most assays were being done in 96-well microplates, which by their very nature had a large footprint and required equally large processing stations,” recounted Tom Ackrill, business development manager of ITL Group, an Ashford, U.K.-based full-service subcontract company in the medical and diagnostic sector providing in vitro diagnostics, point-of-care devices, cancer staging and treatment, surgical robots, and next-generation DNA sequencing. (The company also has a growing presence stateside in Virginia.) “Modern technology has seen assays move from this format down onto single sample chips, and then gradually seems to be moving back towards multiplex assays—multiple tests being performed on a single sample to get the most ‘bang for your buck’ from a single droplet of blood, serum, urine, etc. This, combined with new focus areas such as next-generation sequencing (NGS), antimicrobial stewardship, and a re-emergence of infectious diseases such as Ebola or Plague—plus the nature of caring for an aging population—is pushing toward more rapid bedside tests.”
Speedy diagnostics aren’t just a reaction to society’s collective impatience, however. As Ackrill mentioned, technologies for prompt diagnosis are crucial in order to respond to antimicrobial resistance (AMR) and troublesome emerging infectious diseases. More and more clinicians are encountering pathogen resistance to antibiotic therapies, and these must be diagnosed nearly instantly to protect patients. As is the case with most any blood-borne disease (but more urgent with those like Ebola or Plague) the sooner the diagnosis, the more effective the treatment will be. Firms developing IVD equipment understand this urgency, and constantly push to lower the turnaround time from test to actionable results.
“In infectious disease diagnostics, the ability to deliver clinically actionable information rapidly is critical,” explained Snyder. “Specifically, the rapid identification of infection-causing pathogens directly from patient specimens—rather than from more time-consuming pathogen culturing methods—is an active area of innovation. Similarly, antimicrobial susceptibility testing is a time bottleneck ripe for disruptive innovation. Application of faster and cheaper NGS technology to identify pathogen drug resistance is an area of active development to address this bottleneck.”
“To allow for earlier and more effective therapeutic intervention for infections, many emerging companies are actively developing molecular diagnostic PoC devices that allow for CLIA-waived clinical sample processing and rapid high sensitivity target detection,” Snyder continued. “And as AMR continues to be a pressing unmet clinical dilemma, host immune system status and mobilization is beginning to be recognized as a fertile area of translational research that may lead to novel diagnostic tests to predict infection outcomes—especially in the management of sepsis.”
Go with the Flow
The Coulter principle states that particles pulled through an orifice (concurrent with an electric current) create an impedance change proportional to the volume of the particle traveling through the orifice. The “pulse” in impedance arises from electrolyte displacement caused by the particle. A “Coulter counter” is an apparatus to count and size particles suspended in electrolytes, and has found significant commercial success in hematology to count and size cells making up whole blood. Flow cytometry has its roots in the Coulter principle and is engaged in order to count and sort cells, detect biomarkers, and engineer proteins. In the clinical environment, a flow cytometer is routinely used to diagnosis health disorders, particularly blood cancers.
In case anyone is curious about the sudden science lesson, the patent for the first impedance-based flow cytometer (then called the Coulter counter) was issued to Wallace H. Coulter, who together with his brother Joseph founded Coulter Corporation in 1953. Coulter Corporation was acquired by Beckman Instruments in 1997, establishing Beckman Coulter Inc. (which itself is comprised of two businesses, Life Sciences and Diagnostics, and both divisions reside under Danaher). Among other offerings, the Beckman Coulter Life Sciences business is a manufacturer of clinical and research flow instruments.
“The flow cytometry business continues a strong focus on expanding our offering of clinical products and services,” explained Koksch. “Cellular flow assays are providing very relevant and complementary information to molecular diagnostics assays, routine lab tests, and imaging methods. The advantages of flow assays include a rapid turnaround time, multiplexing capabilities to get more information from our small and precious samples, and analysis of functional cell information.”
Flow cytometry has certainly come a long way since Wallace Coulter’s initial system—modern flow cytometers can analyze thousands of particles a second in real-time. They can also be configured as cell sorters by linking the cytometer’s analytical capability to a sorting device. This enables active separation and isolation of particles with specified optical parameters on a cell-by-cell basis.
Flow cytometry has also become more widely utilized in clinical trials as well as research. New reagents for applications in drug discovery and diagnostics continue to be developed, and the rising prevalence of AIDS and cancer has provoked flow cytometry’s use in scientific research. The technology has even been used to support stem cell research.
“Historically, flow cytometric methods had been widely used to add valuable cellular information to diagnostics and monitoring of treatment approaches for patients suffering from HIV and leukemia and lymphoma, including stem cell transplantations,” Koksch added. “In recent years, we recognize a growing interest in the use of flow methods to assess various aspects of the immune system in the context of cancer, infectious, and autoimmune diseases. While the current focus for these disease states is still on trials and clinical research studies to prove the clinical utility of potential new clinical assays, published results from various studies are very promising.”
Market Prognosis
According to a 2017 EvaluateMedTech report, worldwide IVD sales are expected to reach $70 billion by 2022. The market was valued at about $60.5 billion in 2016, according to the American Association for Clinical Chemistry (AACC). Medical device companies have been paying close attention to the market, and several large, recent M&A transactions have fortified their IVD portfolios. Danaher Corporation completed its $4 billion purchase of Cepheid in late 2016, adding a suite of easy-to-use molecular diagnostic systems and tests, including Cepheid’s GeneXpert systems for rapid genetic testing. Abbott Laboratories also closed its $5.3 billion acquisition of Alere in early October after over 18 months of tumultuous negotiations, adding a portfolio of rapid, PoC tests to the company’s already healthy diagnostic offerings. Despite some volatility in the industry due to changing IVD testing paradigms and reimbursement constraints, companies already established in IVD foresee the market to blossom as patients seek solutions for earlier, more rapid diagnosis.
“We expect the market to continue advancing in technologies that speed the process of identifying diseases and the drugs they are susceptible to,” predicted Snyder. “Customers will seek companies that can provide more holistic solutions for rapidly detecting and monitoring infectious pathogens, where they will have multiple platforms to follow a patient from screening through definitive diagnosis and susceptibility testing.”
The rise of personalized IVD instruments puts a great deal of pressure on suppliers servicing the industry as well. IVD manufacturers actively seek support for new product introductions from their supply chain partners. OEMs will continue to rely on their partners for expertise in areas such as fluid handling and processing, as well as medical material selection.
“With IVD suppliers shipping their products globally, we anticipate suppliers will be required to support their products locally,” stated Colin Rademacher, area sales manager, microfluidics for Bürkert Fluid Control Systems. “OEMs want to work with suppliers who speak the local language and support the products in a region with local support, if need be.”
“As the cost for the new instruments reduces, the trend will keep shifting to the genomic-based personalized IVD instruments,” added Occhiato. “Advancements in handling the large amount of data fuels this trend. We will continue to meet challenges in the fluid handling and processing.”
Further, the continued drive to develop a complete IVD solution will most likely require some degree of customization, particularly in the development of technologies like PoC cartridges. Specialized designers and engineers must have the knowledge in assay development, microfluidics, and systems engineering in order to develop a product from top to bottom. (For more information on this, read “Designing Diagnostic Consumables: Custom or Off-the-Shelf?” a column featured in last month’s issue of MPO.)
“In the IVD industry, there are literally no ‘one-size-fits-all’ solutions,” said Rowan. “Each and every device and test is required to meet different and specific criteria. Every material ranging from a particular reagent solution to filtration and membranous collection materials must be produced according to the testing device to achieve the best performance and most accurate and reliable results. The degree of customization needed in the supply chain to meet industry standards can be challenging. The supply chain plays a significant role in ensuring IVD product manufacturing is conducted without interruption. Companies with expertise in production of specific, varied, and custom microporous membranes are positioned for continued growth due to their agility.”
The flow cytometry market is also expected to grow due to medical and technological advancements in cytometry products, as well as the aforementioned new reagents and cytometry’s adoption in the research landscape. According to Future Market Insights, the segment is expected to reach a high market valuation of $1.8 billion by 2022, with a CAGR of 7.2 percent. Cytometry’s proliferation will also benefit from IVD testing’s shift from traditional laboratories to clinics.
“Flow cytometry as an example of a rapidly evolving IVD technology will serve the need of ease of use, supporting the migration of testing from highly specialized labs to routine clinical testing laboratories and getting diagnostics closer to our patients,” forecasted Koksch. “Overall, the IVD market will gradually evolve from a ‘detect when broken’ to a ‘monitor and prevent’ usage of new IVD solutions.”
According to the AACC, the specific IVD areas of noninvasive prenatal testing, next-generation sequencing, liquid biopsy, and circulating tumor cell (CTC) tests are predicted to explode in the next five years. Statistics from Kalorama Information support this trend—the firm projects NGS test sales to grow from $250 million to $800 million, and CTC tests to rise from 100 million to 300 million. However, the firm also predicts a slowdown in the IVD industry as compared to the previous decade. The industry has previously grown at a pace of 5 percent to 6 percent according to Kalorama, and may slow to about 4 percent each year.
“Patients are starting to expect benefits from all the investments seen over the years and public news stories about technologies promising sci-fi levels of innovation within the next few years,” concluded Ackrill. “Biomarkers also have the potential to play a big role moving forward—ELISA will probably still remain as a dominant force (even if it is antiquated technology) because it is fast, cheap, and effective. The market moving forward will probably involve the handling of more data (and more patient-specific data at that), which then brings the challenges of how to handle, store, and manage that information safely.”
As a result of medicine’s push toward minimally invasive procedures, an alternative, far less painful biopsy method has emerged. Liquid biopsy enables doctors to discover a wealth of information about a tumor through a blood sample (i.e., liquid), rather than a surgical procedure. Fragments of DNA deposited into the bloodstream by tumors have shown success in non-invasive screening for early-stage cancers, monitoring cancer treatment response, and explaining why some cancers resist therapies.
“Procedures of the traditional tissue biopsy are painful, associated with complications such as potential infections, and often available only in central hospitals which require patients to travel long distances,” former MIT postdoc Mohammad Qasaimeh, who was involved in a study in which a microfluidic technique was used to capture and count circulating plasma cells from small samples of blood to diagnose and track multiple myeloma progression, said in a press release. “Capturing plasma cells from blood samples can serve as a liquid biopsy, which can be performed in clinics as often as required, and serve as a diagnostic and prognostic test during and after chemotherapy treatment. Moreover, captured cells can be used for drug testing and thus serve as a tool for personalized medicine.”
This in vitro diagnostic (IVD) method has the potential to transform cancer care, but it’s still relatively early in development. Taking and testing tissue samples still remains the standard, and a number of questions would need to be addressed in clinical trials before making liquid biopsy common practice: How much does test accuracy vary among tumor types and cancer stages? Is the sampling representative of all the genetic clones in a tumor, or is there bias toward particular sub-regions? Until these are answered, liquid biopsy will remain in the clinical research stage.
Established IVD technologies and devices are implemented in either clinical or over-the-counter environments to examine biological samples like blood, saliva, urine, or tissue collected from the body. These devices include chemicals and reagents, analytical equipment, disposables (cartridges or test strips), and software supporting the analytical processes in the lab.
“In the IVD industry, there is growing interest in increasing the portfolio of complete clinical solutions,” illuminated Mario Koksch M.D., Ph.D., MBA, vice president and general manager, Flow Cytometry Business Unit of Indianapolis, Ind.-based Beckman Coulter Life Sciences, part of Danaher Corporation. Dr. Koksch is responsible for the global flow cytometry business, strategy, and manufacturing centers in Marseille, France; Bangalore, India; and Suzhou, China. “A complete clinical solution includes reagents, controls, calibrators, an automated sample prep, and automated data analysis approach. These complete solutions allow for higher quality results in a broader range of labs in less time, which makes initial diagnostics of disease states, monitoring the disease’s progress, and selecting the most appropriate treatment options for individual patients more straightforward, rational, and objective.”
The IVD market is currently undergoing a transformation. Large instruments in central labs are seeing migration to tabletop and hand-held devices, particularly in areas like next-generation sequencing, molecular diagnostics, and point-of-care (PoC). IVD testing has been migrating from the hospital to the physician’s office, and in some cases, to the home. This trend has resulted in a degree of industry consolidation, evidenced by a few significant mergers and acquisitions over the past year. There is also strong pressure to control costs because reimbursement can be challenging for IVD technologies. Because of this and other factors, IVD labs must be incredibly efficient to keep costs down.
“We see a number of increasing challenges facing IVD laboratories,” commented Christine Snyder, senior director of strategic marketing for Diagnostic Systems at Becton, Dickinson and Company (BD), a Franklin Lakes, N.J.-based firm that develops, manufactures, and sells medical supplies, devices, laboratory equipment, and diagnostic products worldwide. “The changing reimbursement landscape, shortage of qualified laboratory technologists, and ongoing consolidation of laboratories means larger organizations are serving more customers with fewer employees. With that, our customers continue to focus on providing fast and accurate test results with greater efficiency, which means we are seeing the widespread adoption of laboratory automation and informatics platforms, products and services. Increasing use of informatics technology helps our customers better manage laboratory information flow and can improve communication between laboratories and physicians.”
Instant Interpretation
The modern world craves immediate results. The medical extension of this need is realized in the impetus for technology to diagnose diseases at the PoC, rather than using traditional lab-based tests. PoC IVD devices fit the bill for this fast-paced attitude because they are usually portable, relatively simple to operate, and the instruments return results rapidly.
Lab-based tests require an order to draw blood, followed by the patient’s trip to the draw site. The specimen is then sent for analysis to the lab, after which the result is finally communicated to the clinician, then finally to the patient or caregiver. Despite labs’ attempts to minimize this turnaround time, it can sometimes take a day or more to receive results. PoC diagnostic systems shortcut the steps of collecting and shipping a specimen; test results can be achieved on the spot in the doctor’s office or at home.
“In the past, in vitro diagnostics were only available in the clinical setting,” recalled Ted Rowan, regional sales manager of Boyd Technologies, a Lee, Mass.-based international supplier and precision converter of advanced flexible materials for the medical device and life sciences markets. “Advances in technology have made it possible to take many tests at home, often with nearly instant results. Home tests are no less accurate, more convenient, and easily performed by the end user. PoC systems are generally membrane-based test strips enclosed inside a plastic cassette. These patient self-tests can detect changes in disease pathophysiology at the earliest onset, empowering patients to take better control of their illnesses.”
“Existing and startup companies manufacturing PoC instruments and kits are advancing and growing rapidly,” observed Craig Occhiato, microfluidics team manager for Bürkert Fluid Control Systems, a Charlotte, N.C.-based manufacturer of measurement and control systems for liquids and gases for the medical device industry. “This is changing the convenience, time, and cost of the IVD landscape—moving it from the large lab and hospital lab to a doctor’s office, patient room, emergency medicine setting, or even a third world country.”
PoC diagnostic technologies generally fall into two categories. The first are small handheld devices with accompanying test strips or cartridges that offer qualitative or quantitative analysis, for example a glucose biosensor fingerstick or a small cartridge device for blood gas analysis. Larger bench-top PoC devices are essentially smaller, less complex versions of laboratory instruments, for example critical care analyzers or small hematology and immunology analyzers.
“When ITL first began work in the field over 40 years ago, most assays were being done in 96-well microplates, which by their very nature had a large footprint and required equally large processing stations,” recounted Tom Ackrill, business development manager of ITL Group, an Ashford, U.K.-based full-service subcontract company in the medical and diagnostic sector providing in vitro diagnostics, point-of-care devices, cancer staging and treatment, surgical robots, and next-generation DNA sequencing. (The company also has a growing presence stateside in Virginia.) “Modern technology has seen assays move from this format down onto single sample chips, and then gradually seems to be moving back towards multiplex assays—multiple tests being performed on a single sample to get the most ‘bang for your buck’ from a single droplet of blood, serum, urine, etc. This, combined with new focus areas such as next-generation sequencing (NGS), antimicrobial stewardship, and a re-emergence of infectious diseases such as Ebola or Plague—plus the nature of caring for an aging population—is pushing toward more rapid bedside tests.”
Speedy diagnostics aren’t just a reaction to society’s collective impatience, however. As Ackrill mentioned, technologies for prompt diagnosis are crucial in order to respond to antimicrobial resistance (AMR) and troublesome emerging infectious diseases. More and more clinicians are encountering pathogen resistance to antibiotic therapies, and these must be diagnosed nearly instantly to protect patients. As is the case with most any blood-borne disease (but more urgent with those like Ebola or Plague) the sooner the diagnosis, the more effective the treatment will be. Firms developing IVD equipment understand this urgency, and constantly push to lower the turnaround time from test to actionable results.
“In infectious disease diagnostics, the ability to deliver clinically actionable information rapidly is critical,” explained Snyder. “Specifically, the rapid identification of infection-causing pathogens directly from patient specimens—rather than from more time-consuming pathogen culturing methods—is an active area of innovation. Similarly, antimicrobial susceptibility testing is a time bottleneck ripe for disruptive innovation. Application of faster and cheaper NGS technology to identify pathogen drug resistance is an area of active development to address this bottleneck.”
“To allow for earlier and more effective therapeutic intervention for infections, many emerging companies are actively developing molecular diagnostic PoC devices that allow for CLIA-waived clinical sample processing and rapid high sensitivity target detection,” Snyder continued. “And as AMR continues to be a pressing unmet clinical dilemma, host immune system status and mobilization is beginning to be recognized as a fertile area of translational research that may lead to novel diagnostic tests to predict infection outcomes—especially in the management of sepsis.”
Go with the Flow
The Coulter principle states that particles pulled through an orifice (concurrent with an electric current) create an impedance change proportional to the volume of the particle traveling through the orifice. The “pulse” in impedance arises from electrolyte displacement caused by the particle. A “Coulter counter” is an apparatus to count and size particles suspended in electrolytes, and has found significant commercial success in hematology to count and size cells making up whole blood. Flow cytometry has its roots in the Coulter principle and is engaged in order to count and sort cells, detect biomarkers, and engineer proteins. In the clinical environment, a flow cytometer is routinely used to diagnosis health disorders, particularly blood cancers.
In case anyone is curious about the sudden science lesson, the patent for the first impedance-based flow cytometer (then called the Coulter counter) was issued to Wallace H. Coulter, who together with his brother Joseph founded Coulter Corporation in 1953. Coulter Corporation was acquired by Beckman Instruments in 1997, establishing Beckman Coulter Inc. (which itself is comprised of two businesses, Life Sciences and Diagnostics, and both divisions reside under Danaher). Among other offerings, the Beckman Coulter Life Sciences business is a manufacturer of clinical and research flow instruments.
“The flow cytometry business continues a strong focus on expanding our offering of clinical products and services,” explained Koksch. “Cellular flow assays are providing very relevant and complementary information to molecular diagnostics assays, routine lab tests, and imaging methods. The advantages of flow assays include a rapid turnaround time, multiplexing capabilities to get more information from our small and precious samples, and analysis of functional cell information.”
Flow cytometry has certainly come a long way since Wallace Coulter’s initial system—modern flow cytometers can analyze thousands of particles a second in real-time. They can also be configured as cell sorters by linking the cytometer’s analytical capability to a sorting device. This enables active separation and isolation of particles with specified optical parameters on a cell-by-cell basis.
Flow cytometry has also become more widely utilized in clinical trials as well as research. New reagents for applications in drug discovery and diagnostics continue to be developed, and the rising prevalence of AIDS and cancer has provoked flow cytometry’s use in scientific research. The technology has even been used to support stem cell research.
“Historically, flow cytometric methods had been widely used to add valuable cellular information to diagnostics and monitoring of treatment approaches for patients suffering from HIV and leukemia and lymphoma, including stem cell transplantations,” Koksch added. “In recent years, we recognize a growing interest in the use of flow methods to assess various aspects of the immune system in the context of cancer, infectious, and autoimmune diseases. While the current focus for these disease states is still on trials and clinical research studies to prove the clinical utility of potential new clinical assays, published results from various studies are very promising.”
Market Prognosis
According to a 2017 EvaluateMedTech report, worldwide IVD sales are expected to reach $70 billion by 2022. The market was valued at about $60.5 billion in 2016, according to the American Association for Clinical Chemistry (AACC). Medical device companies have been paying close attention to the market, and several large, recent M&A transactions have fortified their IVD portfolios. Danaher Corporation completed its $4 billion purchase of Cepheid in late 2016, adding a suite of easy-to-use molecular diagnostic systems and tests, including Cepheid’s GeneXpert systems for rapid genetic testing. Abbott Laboratories also closed its $5.3 billion acquisition of Alere in early October after over 18 months of tumultuous negotiations, adding a portfolio of rapid, PoC tests to the company’s already healthy diagnostic offerings. Despite some volatility in the industry due to changing IVD testing paradigms and reimbursement constraints, companies already established in IVD foresee the market to blossom as patients seek solutions for earlier, more rapid diagnosis.
“We expect the market to continue advancing in technologies that speed the process of identifying diseases and the drugs they are susceptible to,” predicted Snyder. “Customers will seek companies that can provide more holistic solutions for rapidly detecting and monitoring infectious pathogens, where they will have multiple platforms to follow a patient from screening through definitive diagnosis and susceptibility testing.”
The rise of personalized IVD instruments puts a great deal of pressure on suppliers servicing the industry as well. IVD manufacturers actively seek support for new product introductions from their supply chain partners. OEMs will continue to rely on their partners for expertise in areas such as fluid handling and processing, as well as medical material selection.
“With IVD suppliers shipping their products globally, we anticipate suppliers will be required to support their products locally,” stated Colin Rademacher, area sales manager, microfluidics for Bürkert Fluid Control Systems. “OEMs want to work with suppliers who speak the local language and support the products in a region with local support, if need be.”
“As the cost for the new instruments reduces, the trend will keep shifting to the genomic-based personalized IVD instruments,” added Occhiato. “Advancements in handling the large amount of data fuels this trend. We will continue to meet challenges in the fluid handling and processing.”
Further, the continued drive to develop a complete IVD solution will most likely require some degree of customization, particularly in the development of technologies like PoC cartridges. Specialized designers and engineers must have the knowledge in assay development, microfluidics, and systems engineering in order to develop a product from top to bottom. (For more information on this, read “Designing Diagnostic Consumables: Custom or Off-the-Shelf?” a column featured in last month’s issue of MPO.)
“In the IVD industry, there are literally no ‘one-size-fits-all’ solutions,” said Rowan. “Each and every device and test is required to meet different and specific criteria. Every material ranging from a particular reagent solution to filtration and membranous collection materials must be produced according to the testing device to achieve the best performance and most accurate and reliable results. The degree of customization needed in the supply chain to meet industry standards can be challenging. The supply chain plays a significant role in ensuring IVD product manufacturing is conducted without interruption. Companies with expertise in production of specific, varied, and custom microporous membranes are positioned for continued growth due to their agility.”
The flow cytometry market is also expected to grow due to medical and technological advancements in cytometry products, as well as the aforementioned new reagents and cytometry’s adoption in the research landscape. According to Future Market Insights, the segment is expected to reach a high market valuation of $1.8 billion by 2022, with a CAGR of 7.2 percent. Cytometry’s proliferation will also benefit from IVD testing’s shift from traditional laboratories to clinics.
“Flow cytometry as an example of a rapidly evolving IVD technology will serve the need of ease of use, supporting the migration of testing from highly specialized labs to routine clinical testing laboratories and getting diagnostics closer to our patients,” forecasted Koksch. “Overall, the IVD market will gradually evolve from a ‘detect when broken’ to a ‘monitor and prevent’ usage of new IVD solutions.”
According to the AACC, the specific IVD areas of noninvasive prenatal testing, next-generation sequencing, liquid biopsy, and circulating tumor cell (CTC) tests are predicted to explode in the next five years. Statistics from Kalorama Information support this trend—the firm projects NGS test sales to grow from $250 million to $800 million, and CTC tests to rise from 100 million to 300 million. However, the firm also predicts a slowdown in the IVD industry as compared to the previous decade. The industry has previously grown at a pace of 5 percent to 6 percent according to Kalorama, and may slow to about 4 percent each year.
“Patients are starting to expect benefits from all the investments seen over the years and public news stories about technologies promising sci-fi levels of innovation within the next few years,” concluded Ackrill. “Biomarkers also have the potential to play a big role moving forward—ELISA will probably still remain as a dominant force (even if it is antiquated technology) because it is fast, cheap, and effective. The market moving forward will probably involve the handling of more data (and more patient-specific data at that), which then brings the challenges of how to handle, store, and manage that information safely.”