Maria Shepherd, President and Founder, Medi-Vantage06.06.17
Boston, Mass., was the place to be in the first week of May 2017 for people interested in the business of medtech. The World Medical Innovation Forum kicked off the week for a three-day meeting that maintained a focus on cardiovascular disease and technology. The MassMEDIC Annual Conference followed and was co-located with the BIOMEDevice Boston event—a medtech product development conference focused on the technologies that support medical device development in our space.
Why This Is Important
The message heard at all three meetings was that the business of medical devices is growing and the opportunity for innovation is critically important to grow the industry. Omar Ishrak, president and CEO of Medtronic plc, explained it best when he described the innovations coming out of his company, especially those that are revitalizing the cardiac pacemaker industry.
“We’ve taken a commodity product and created a new platform, called Micra, of very small, lead-free pacemakers,” said Ishrak. “Medtronic will be able to expand upon this new platform to create multiple iterations of pacemakers for years
to come.”
Micra is one of the first products developed from Medtronic’s “deep miniaturization” project.
Multiple Opportunities to Recreate Medical Device Commodity Products
According to a report,1 this product improvement comes in the nick of time to dominate market growth in cardiac pacemakers. Global sales for cardiac pacemakers was estimated at $7.5 billion in 2015 and is forecasted to grow to almost $12 billion by the end of 2021 (Chart 1).

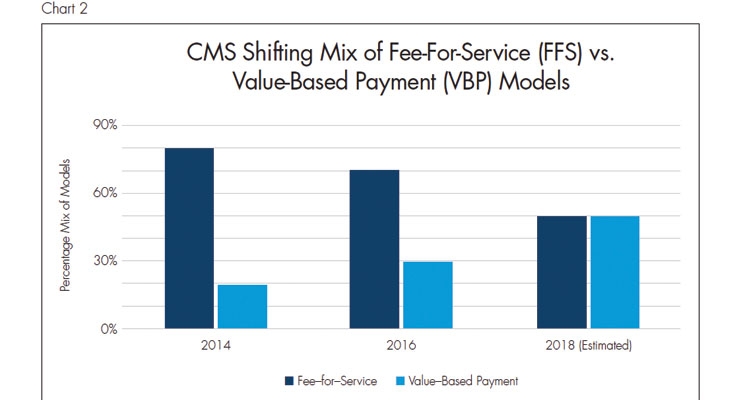
Pacemakers aren’t the only medtech product platform that can benefit from miniaturization. It is important to recognize, however, that making medtech products smaller can’t be the only design goal. The lesson learned from Micra is that clinical outcomes must be improved while reducing cost to the healthcare organization. Micra accomplishes this goal by reducing complications such as device dislodgement or infections.2 Reducing cost to healthcare organizations is critical because, as discussed at a panel at the MassMEDIC 21st Annual Conference in Boston, payers are shifting to value-based payment models3 (Chart 2). Although it has taken longer than originally intended, this means that healthcare providers will continue to take on more risk.
Expect More Innovation to Change the Medtech Landscape
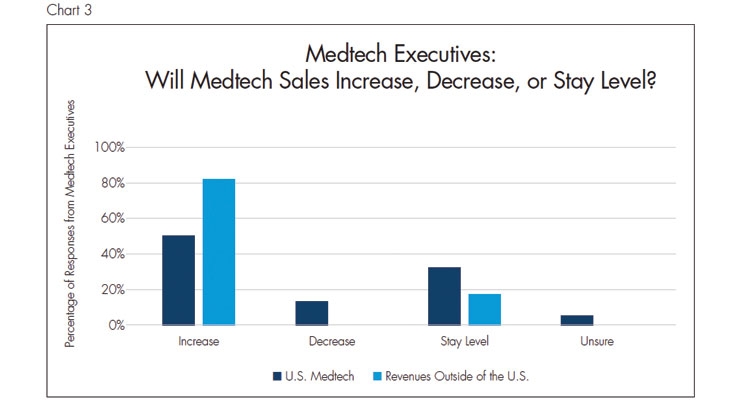
All three meetings discussed the rate of innovation in medical devices, but the BIOMEDevice meeting focused on new technologies and how product development teams can keep up with the rapid changes in the medtech market. Data from the most recent MassMEDIC Medtech Industry and Innovation Study4 (n=123) was included in a presentation at BIOMEDevice. This study stated that the majority of medical technology executives surveyed indicated that medtech sales will increase and that product development teams need to focus on innovation.
Conclusion and Recommendations
Innovation is critically important in medtech, where we have barely scratched the surface of what medical devices can do to improve patient outcomes and reduce the cost of healthcare. Which of your commodity products can be improved and elevated into a new platform of innovation?
References
Maria Shepherd has more than 20 years of leadership experience in medical device/life-science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical—where she boosted the company valuation prior to its acquisition by Covidien/Medtronic—director of marketing for Philips Medical, and senior management roles at Boston Scientific Corp., she founded Medi-Vantage. Medi-Vantage provides marketing and business strategy and innovation research for the medical device industry. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught marketing and product development courses and is a member of the Aligo Medtech Investment Committee (www.msbiv.com). She can be reached at 855-343-3100 ext. 102 or at mshepherd@medi-vantage.com.
Why This Is Important
The message heard at all three meetings was that the business of medical devices is growing and the opportunity for innovation is critically important to grow the industry. Omar Ishrak, president and CEO of Medtronic plc, explained it best when he described the innovations coming out of his company, especially those that are revitalizing the cardiac pacemaker industry.
“We’ve taken a commodity product and created a new platform, called Micra, of very small, lead-free pacemakers,” said Ishrak. “Medtronic will be able to expand upon this new platform to create multiple iterations of pacemakers for years
to come.”
Micra is one of the first products developed from Medtronic’s “deep miniaturization” project.
Multiple Opportunities to Recreate Medical Device Commodity Products
According to a report,1 this product improvement comes in the nick of time to dominate market growth in cardiac pacemakers. Global sales for cardiac pacemakers was estimated at $7.5 billion in 2015 and is forecasted to grow to almost $12 billion by the end of 2021 (Chart 1).

Pacemakers aren’t the only medtech product platform that can benefit from miniaturization. It is important to recognize, however, that making medtech products smaller can’t be the only design goal. The lesson learned from Micra is that clinical outcomes must be improved while reducing cost to the healthcare organization. Micra accomplishes this goal by reducing complications such as device dislodgement or infections.2 Reducing cost to healthcare organizations is critical because, as discussed at a panel at the MassMEDIC 21st Annual Conference in Boston, payers are shifting to value-based payment models3 (Chart 2). Although it has taken longer than originally intended, this means that healthcare providers will continue to take on more risk.

Expect More Innovation to Change the Medtech Landscape
All three meetings discussed the rate of innovation in medical devices, but the BIOMEDevice meeting focused on new technologies and how product development teams can keep up with the rapid changes in the medtech market. Data from the most recent MassMEDIC Medtech Industry and Innovation Study4 (n=123) was included in a presentation at BIOMEDevice. This study stated that the majority of medical technology executives surveyed indicated that medtech sales will increase and that product development teams need to focus on innovation.

Conclusion and Recommendations
Innovation is critically important in medtech, where we have barely scratched the surface of what medical devices can do to improve patient outcomes and reduce the cost of healthcare. Which of your commodity products can be improved and elevated into a new platform of innovation?
References
- http://bit.ly/mpo061701
- http://bit.ly/mpo061702
- http://bit.ly/mpo061703 [PDF]
- http://bit.ly/mpo061704 [PDF]
Maria Shepherd has more than 20 years of leadership experience in medical device/life-science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical—where she boosted the company valuation prior to its acquisition by Covidien/Medtronic—director of marketing for Philips Medical, and senior management roles at Boston Scientific Corp., she founded Medi-Vantage. Medi-Vantage provides marketing and business strategy and innovation research for the medical device industry. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught marketing and product development courses and is a member of the Aligo Medtech Investment Committee (www.msbiv.com). She can be reached at 855-343-3100 ext. 102 or at mshepherd@medi-vantage.com.