Sam Brusco, Associate Editor11.08.16
It’s difficult not to imagine Star Trek’s replicator whenever the subject of 3D printing is brought up. To explain for the less nerdy MPO readers: The replicator is a boxlike machine capable of creating and recycling objects. It can reconstitute matter to produce necessary items—including food, medication, and spare parts—out of pure energy.
Obviously, 3D printers don’t actually rearrange subatomic particles to create material ex nihilo (out of nothing) per the show’s canon. But one has to admit the replicator’s physical resemblance to modern 3D printers is startlingly uncanny. The types of items being printed also match up surprisingly well; early in the show’s run, the replicator was used mainly for food and water provision, the former of which some researchers have been able to accomplish. (The food doesn’t look all that appetizing, unfortunately—hopefully researchers are working on that.) In later episodes, the replicator began making both medicine and spare parts, which 3D printers have begun producing this year. Aprecia Pharmaceuticals won U.S. Food and Drug Administration (FDA) approval for the first 3D-printed drug in March. Perhaps the most intriguing Star Trek parallel that has moved outside the realm of fiction occurred in September, when GE Aviation introduced the first ever 3D-printed aircraft engine parts.
3D printing’s potential for medical applications is staggering, and is slowly but surely evolving beyond its use as merely a rapid prototyping resource. “It’s hard to believe 3D printing in the medical field started in the early 2000s with the development of custom dental implants,” mused Patty Nichols, second vice president of medical technology underwriting for New York, N.Y.-based Travelers. “3D printers are now being used to create hearing aids, contact lenses, and prosthetics made to an individual patient’s exact body shape and contours, often at a fraction of the cost of a conventional medical device.”
“We have seen significant increases in demand for 3D printing in the medical space,” noted Michael Gaisford, director of marketing, medical solutions, for Eden Prairie, Minn.-based Stratasys Ltd. “It is one of our highest growth segments. The primary application we see driving the growth is the printing of medical models that can be used for patient-specific surgical planning at hospitals, anatomical simulators to train physicians on new procedures and devices, and replace animals and cadavers for device testing for verification and validation.”
3D printing’s unique ability to provide the tools necessary to customize and test devices, as well as plan procedures, becomes remarkably timely in the context of today’s healthcare environment, which is moving inexorably toward individualized treatments that are tailored to a patient’s specific needs. Because patient CT scans can be imported into design software as a reference model, the manufacture of “patient-specific” devices has become a viable business.
“Our suite of backbone technology allows medical companies and clinicians to go from scan data all the way through production in a certified, cleared environment,” said Bryan Crutchfield, vice president and general manager, North America, for Leuven, Belgium-based Materialise. “In some cases, we are providing solutions that take patient specific scan data; plan the treatment interactively with the clinician; [and] produce the surgical tools, anatomical models, and some implants. In other areas, we are providing the software and population-driven technical services that medical device companies use to target patient populations. Additionally, through our software and anatomical models, we are assisting clinicians in the preoperative planning and approaches to improve outcomes.”
Crutchfield continued, saying, “New and innovative 3D printed medical solutions are continuously being developed that allow for treatment of previously untreatable conditions. Recent innovations include 3D printed pharmaceuticals, disposable surgical tools, tissue scaffolds for regenerative medicine, and resorbable devices, such as the tracheal splint at the University of Michigan.”
Crutchfield is of course referring to the polycaprolactone splint produced in February 2012 for newborn Kaiba Gionfriddo, whose bronchus collapsed nearly every day, blocking the flow of air to his lungs. Created directly from a CT scan of Kaiba’s trachea, Materialise’s software helped University of Michigan researchers to design and size the device. It then underwent emergency U.S. FDA clearance and was implanted into Kaiba, who flourishes today without “another episode of turning blue,” as his mother April Gionfriddo recounted.
And it’s not just what 3D printers are churning out that’s advancing—the capability of 3D printers to further customize products, mix materials, and incorporate novel materials is becoming quite impressive as well.
“One of the most significant advancements is the introduction of full-color 3D printing with the ability to match soft and hard texture,” said Gaisford. “In the past, we could print with three materials simultaneously, which meant that if one wanted to include soft materials in a model, there were only two color possibilities. Our new model can print six materials, so there is no tradeoff in color and texture. That means we can print realistic, dynamic simulation models that physicians can perform procedures on that also look realistic, or include important color differentiation to guide learning.”
David Somers, senior industry analyst for Yardley, Pa.-based strategic advisory firm Axendia Inc., described an ongoing effort concerning the licensing of, and service level agreements for, 3D printed devices. “It would be possible to set up scenarios where the device company and/or brand owner has the rights over the intellectual property, design, and additive manufacturing strategy,” he said. “But the device itself is printed on demand at the point of use (provider). In this scenario, the brand owner would be responsible for functional and operational specifications and applicable compliance activities. This approach would make it possible to have a pay-per-use scenario. The FDA is still considering the ramifications of this kind of arrangement.”
Moving Beyond Prototyping
Rapid prototyping isn’t the only practical song additive manufacturing is singing. According to the FDA website, as of December 2015, the agency had cleared more than 85 3D printed medical devices. The agency has cleared a number of 3D printed devices this year—some of which were implantable—which is particularly impressive as these devices are often subject to a more stringent review process.
Another large stride for the technology was made in May of this year, when the FDA issued a long-overdue Draft Guidance (DG) for 3D printing of medical devices entitled “Technical Considerations for Additive Manufactured Devices.” Granted, the guidance is still undergoing review and it’s not quite known when these recommendations will be finalized. But if medical device manufacturers hadn’t caught the 3D printing bug already, the DG certainly made them aware of its potential—though potential doesn’t seem like a strong enough word to describe the new advancements.
“We see customers using 3D printing for pretty much every step in the value chain,” remarked Gaisford. “Device testing on anatomically accurate models that include pathology is a growing application of interest. Product parts for clinical trials as well as early commercialization are also being 3D printed to delay investment in production tooling. Companies, medical schools, and hospitals are using 3D printing to train physicians on new skills. And of course, hospitals are using 3D models for patient care by printing models of their diseased anatomy to allow physicians to plan, practice, and determine optimal approaches. The versatility of 3D printing is allowing organizations to generate multiple sources of value from a single piece of equipment.”
“There are many applications in medtech beyond prototyping,” explained Crutchfield. “These include surgical guides and tools, resorbable implants, titanium implants, anatomical models, dental wafers, teeth aligners, customized prosthetics, and production process aids like fixtures and tooling. Through our workflow solution partners in the industry, we see millions of these items printed each year.”
Arguably the most exciting—and easily the most “sci-fi”—of potential 3D printing applications is “bio printing,” or on-demand production of living tissue. In this process, stem-cell-containing “bio-inks” are printed into cell patterns layer by layer to create tissue-like structures that can be used for tissue engineering. 3D bio printing has also begun to incorporate biodegradable scaffolds in order to regenerate joints and ligaments.
“Bio printing may prove to be the most disruptive, yet welcome technology of the 21st century,” said Nichols. “Researchers can now fabricate human tissue with 3D printers and a patient’s own DNA. Using biodegradable scaffolds, doctors can print an organ’s framework, then inject it with a patient’s own living cells in the exact location where they are most likely to grow naturally. Because the patient’s own cells are injected into the bio printed material, the risk of rejection is minimized.”
However, useful as on-demand tissues and organs may be, there’s still quite a bit to hash out before these “devices” are adopted. There’s not yet a regulated process for printing biological materials, nor are there many printers (outside of the research and development space) equipped to handle living tissue. Some do exist—inkjet, laser-assisted, and extrusion printers have been used for biological applications. In fact, a startup called BioBots even developed a printer designed exclusively for bio printing last year.

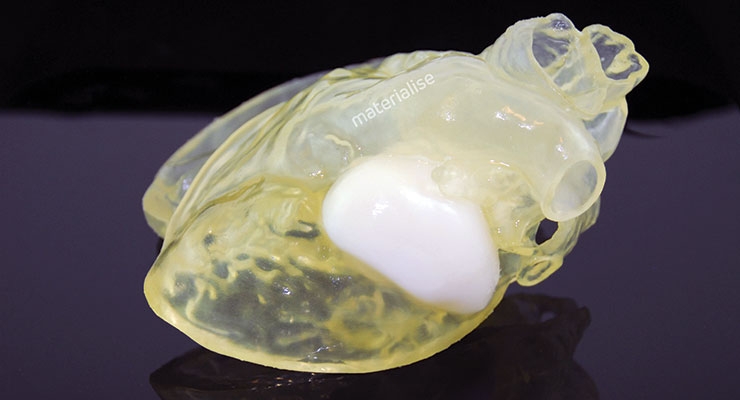
Liver model used for surgical planning of a live donor liver transplant procedure at the Cleveland Clinic. Image courtesy of Stratasys.
However, one medical 3D printing application does exist that device manufacturers might not even be aware of, which commercial 3D printing service providers actually offer: the manufacture of customized anatomical models.
“We believe the medical models application is one of the under-utilized areas when it comes to medical OEM adoption,” said Gaisford. “Hospitals and physicians are leading the charge here, especially for surgical planning. But medical OEMs are just learning about the ways 3D printed models can enhance device testing and physician education beyond existing modalities of human cadavers and animals, which have significant limitations. With cadavers, the parts have been processed and no longer retain living tissue characteristics and it is often difficult to find a cadaver that has the target pathology within it. Animal models only approximate human anatomy and also lack disease. Many medical OEMs have not stayed up to date with developments in materials and printer capabilities that enable 3D printed models to mimic clinical scenarios more accurately than in the past and to match color realism.”
“Our cleared Materialise Mimics software provides clinicians with the ability to plan and print anatomical models for highly complex cases,” said Crutchfield. “These anatomical models and surgical plans allow for a comprehensive diagnosis and treatment plan that shortens procedure times and improves outcomes. However, adoption has been slowed by lack of reimbursement by benefit providers who have yet to catch up with the technology.”
Hurdles to Larger Adoption
As with any emerging power, 3D printing obviously can’t grow unchecked. Though some believe its abilities could be limitless, medical applications for 3D printing still remain in the adolescent stage. This isn’t altogether unexpected, as some medical applications—bio printing being one example—require 3D printing to boldly go where it isn’t quite ready to go just yet. 3D printers will eventually have to adapt to handle multiple materials, biological components, and different sets of software solutions to accomplish what particularly progressive OEMs hope to achieve.
“Right now, materials capability is the greatest hurdle. Customers are asking for materials that are as hard as bone or soft as skin from the same printer,” said Gaisford. “That’s an incredible technical challenge to achieve. It’s something we are working very hard to improve. The other hurdle is the ease of use of going from concept to printed part. For example, to take an MRI/CT to printed part can take multiple software packages and trained expertise. We need to find ways to better streamline that workflow.”
“The capability for ‘individual’ or ‘custom-bespoke’ manufacturing capabilities requires a more comprehensive data collection and tracking for each patient; a process some manufacturers might not want to take responsibility for,” noted Somers. But he also offered a potential solution to this hurdle, suggesting that “instead, intermediary companies can be used within the supply chain—by either medical groups offering a wide range of services to monitor it for their ‘subscribers,’ or even insurance companies, who mostly pay for them.”
Crutchfield also offered advice on the best way to advance 3D printing’s practicality—and it’s no surprise that the brunt of that work lies with the industry’s friendly neighborhood regulatory body.
“Finalization of the FDA Draft Guidance will provide for a stable basis for companies to build from going forward in implementing additive manufacturing solutions in the market,” he suggested.
Somers expressed a similar view on the most practical way to leap over the hurdles of wider medical 3D printing adoption—aptly navigating any regulatory complexities throughout the entire manufacturing process.
“Since the sea change at FDA from ‘rule-based’ to ‘risk-based’ strategies associated with inspection and evaluation criteria, the onus has been shifted to the manufacturers to provide the statistical data and analyses for their products to be reviewed and monitored,” he explained. “I would apply a similar scenario to the 3D manufacturing: if the products pass the ‘benefit-risk’ test and data is provided ‘as requested’ regarding their functional and operational history, there shouldn’t be any interference with proprietary processes by the FDA.”
Further, Crutchfield explained the importance of payers’ roles in the technology’s wider adoption. “Additionally, acceptance of new 3D printing solutions in the surgical guide and anatomical market as reimbursable items by Medicare/Medicaid and benefit providers is needed.”
Though more a byproduct than a hurdle of larger 3D printing adoption, Nichols stressed the importance of cybersecurity—a topic that’s certainly at the front of the minds of those in the medtech industry who have been paying attention to recent claims of vulnerabilities (whether they are founded or not) in St. Jude Medical Inc.’s implantable cardiac devices.
“Privacy and security will be important considerations as patient-specific data will need to be inputted and transmitted safely,” she advised. “No company is immune to a cyber-extortion scheme, virus, or other malware that could affect their manufacturing systems. In addition, verifying the safety and efficacy of the products that are being manufactured using 3D printing in medtech will be imperative.”
What’s Next on the Queue?
It seems strange to ask where the future of 3D printing is headed when the technology is already so futuristic. But those involved in additive manufacturing, whether providing printing services or developing devices, are always looking ahead to stay on top of the fertile ground 3D printing has laid for medtech development.
“I think in the next five years, we will see new materials with greater clearances and certifications for use in production parts,” envisioned Gaisford.
“The medical industry will continue to push the boundaries of additive manufacturing in the future, as the industry drives towards cost-effective, patient-specific solutions that address each unique condition,” predicted Crutchfield. “As we work with our workflow solution partners we see several areas developing in the next five years, such as patient-specific metal implants, resorbable implants, bio printing, and 3D printed pharmacology—just to name a few.”
“The industry is seeing an increase in the number of ‘point of care’ device manufacturing, where 3D printers are now located in the hospital where a patient’s specific part can be manufactured on the spot,” Nichols noted. But she also brought up an important point regarding who is truly “responsible” for a device that is manufactured in this way. “This raises an interesting question from a products liability perspective, as the lines are blurred on who truly is the ‘manufacturer’ of the final medical product—is it the 3D manufacturer, the hospital, or the physician?”
Somers was optimistic in his forecast, saying, “3D printing will continue to move ever upward.” He was also hopeful for additional next-generation technologies that could one day companion products manufactured by 3D printing to develop treatments that healthcare professionals might never have dreamed of. “[3D printing], along with nanotechnology and other biological process therapies, are revolutionizing the medical treatment field, all for the benefit of patients.”
Obviously, 3D printers don’t actually rearrange subatomic particles to create material ex nihilo (out of nothing) per the show’s canon. But one has to admit the replicator’s physical resemblance to modern 3D printers is startlingly uncanny. The types of items being printed also match up surprisingly well; early in the show’s run, the replicator was used mainly for food and water provision, the former of which some researchers have been able to accomplish. (The food doesn’t look all that appetizing, unfortunately—hopefully researchers are working on that.) In later episodes, the replicator began making both medicine and spare parts, which 3D printers have begun producing this year. Aprecia Pharmaceuticals won U.S. Food and Drug Administration (FDA) approval for the first 3D-printed drug in March. Perhaps the most intriguing Star Trek parallel that has moved outside the realm of fiction occurred in September, when GE Aviation introduced the first ever 3D-printed aircraft engine parts.
3D printing’s potential for medical applications is staggering, and is slowly but surely evolving beyond its use as merely a rapid prototyping resource. “It’s hard to believe 3D printing in the medical field started in the early 2000s with the development of custom dental implants,” mused Patty Nichols, second vice president of medical technology underwriting for New York, N.Y.-based Travelers. “3D printers are now being used to create hearing aids, contact lenses, and prosthetics made to an individual patient’s exact body shape and contours, often at a fraction of the cost of a conventional medical device.”
“We have seen significant increases in demand for 3D printing in the medical space,” noted Michael Gaisford, director of marketing, medical solutions, for Eden Prairie, Minn.-based Stratasys Ltd. “It is one of our highest growth segments. The primary application we see driving the growth is the printing of medical models that can be used for patient-specific surgical planning at hospitals, anatomical simulators to train physicians on new procedures and devices, and replace animals and cadavers for device testing for verification and validation.”
3D printing’s unique ability to provide the tools necessary to customize and test devices, as well as plan procedures, becomes remarkably timely in the context of today’s healthcare environment, which is moving inexorably toward individualized treatments that are tailored to a patient’s specific needs. Because patient CT scans can be imported into design software as a reference model, the manufacture of “patient-specific” devices has become a viable business.
“Our suite of backbone technology allows medical companies and clinicians to go from scan data all the way through production in a certified, cleared environment,” said Bryan Crutchfield, vice president and general manager, North America, for Leuven, Belgium-based Materialise. “In some cases, we are providing solutions that take patient specific scan data; plan the treatment interactively with the clinician; [and] produce the surgical tools, anatomical models, and some implants. In other areas, we are providing the software and population-driven technical services that medical device companies use to target patient populations. Additionally, through our software and anatomical models, we are assisting clinicians in the preoperative planning and approaches to improve outcomes.”
Crutchfield continued, saying, “New and innovative 3D printed medical solutions are continuously being developed that allow for treatment of previously untreatable conditions. Recent innovations include 3D printed pharmaceuticals, disposable surgical tools, tissue scaffolds for regenerative medicine, and resorbable devices, such as the tracheal splint at the University of Michigan.”
Crutchfield is of course referring to the polycaprolactone splint produced in February 2012 for newborn Kaiba Gionfriddo, whose bronchus collapsed nearly every day, blocking the flow of air to his lungs. Created directly from a CT scan of Kaiba’s trachea, Materialise’s software helped University of Michigan researchers to design and size the device. It then underwent emergency U.S. FDA clearance and was implanted into Kaiba, who flourishes today without “another episode of turning blue,” as his mother April Gionfriddo recounted.
And it’s not just what 3D printers are churning out that’s advancing—the capability of 3D printers to further customize products, mix materials, and incorporate novel materials is becoming quite impressive as well.
“One of the most significant advancements is the introduction of full-color 3D printing with the ability to match soft and hard texture,” said Gaisford. “In the past, we could print with three materials simultaneously, which meant that if one wanted to include soft materials in a model, there were only two color possibilities. Our new model can print six materials, so there is no tradeoff in color and texture. That means we can print realistic, dynamic simulation models that physicians can perform procedures on that also look realistic, or include important color differentiation to guide learning.”
David Somers, senior industry analyst for Yardley, Pa.-based strategic advisory firm Axendia Inc., described an ongoing effort concerning the licensing of, and service level agreements for, 3D printed devices. “It would be possible to set up scenarios where the device company and/or brand owner has the rights over the intellectual property, design, and additive manufacturing strategy,” he said. “But the device itself is printed on demand at the point of use (provider). In this scenario, the brand owner would be responsible for functional and operational specifications and applicable compliance activities. This approach would make it possible to have a pay-per-use scenario. The FDA is still considering the ramifications of this kind of arrangement.”
Moving Beyond Prototyping
Rapid prototyping isn’t the only practical song additive manufacturing is singing. According to the FDA website, as of December 2015, the agency had cleared more than 85 3D printed medical devices. The agency has cleared a number of 3D printed devices this year—some of which were implantable—which is particularly impressive as these devices are often subject to a more stringent review process.
Another large stride for the technology was made in May of this year, when the FDA issued a long-overdue Draft Guidance (DG) for 3D printing of medical devices entitled “Technical Considerations for Additive Manufactured Devices.” Granted, the guidance is still undergoing review and it’s not quite known when these recommendations will be finalized. But if medical device manufacturers hadn’t caught the 3D printing bug already, the DG certainly made them aware of its potential—though potential doesn’t seem like a strong enough word to describe the new advancements.
“We see customers using 3D printing for pretty much every step in the value chain,” remarked Gaisford. “Device testing on anatomically accurate models that include pathology is a growing application of interest. Product parts for clinical trials as well as early commercialization are also being 3D printed to delay investment in production tooling. Companies, medical schools, and hospitals are using 3D printing to train physicians on new skills. And of course, hospitals are using 3D models for patient care by printing models of their diseased anatomy to allow physicians to plan, practice, and determine optimal approaches. The versatility of 3D printing is allowing organizations to generate multiple sources of value from a single piece of equipment.”
“There are many applications in medtech beyond prototyping,” explained Crutchfield. “These include surgical guides and tools, resorbable implants, titanium implants, anatomical models, dental wafers, teeth aligners, customized prosthetics, and production process aids like fixtures and tooling. Through our workflow solution partners in the industry, we see millions of these items printed each year.”
Arguably the most exciting—and easily the most “sci-fi”—of potential 3D printing applications is “bio printing,” or on-demand production of living tissue. In this process, stem-cell-containing “bio-inks” are printed into cell patterns layer by layer to create tissue-like structures that can be used for tissue engineering. 3D bio printing has also begun to incorporate biodegradable scaffolds in order to regenerate joints and ligaments.
“Bio printing may prove to be the most disruptive, yet welcome technology of the 21st century,” said Nichols. “Researchers can now fabricate human tissue with 3D printers and a patient’s own DNA. Using biodegradable scaffolds, doctors can print an organ’s framework, then inject it with a patient’s own living cells in the exact location where they are most likely to grow naturally. Because the patient’s own cells are injected into the bio printed material, the risk of rejection is minimized.”
However, useful as on-demand tissues and organs may be, there’s still quite a bit to hash out before these “devices” are adopted. There’s not yet a regulated process for printing biological materials, nor are there many printers (outside of the research and development space) equipped to handle living tissue. Some do exist—inkjet, laser-assisted, and extrusion printers have been used for biological applications. In fact, a startup called BioBots even developed a printer designed exclusively for bio printing last year.

Liver model used for surgical planning of a live donor liver transplant procedure at the Cleveland Clinic. Image courtesy of Stratasys.
“We believe the medical models application is one of the under-utilized areas when it comes to medical OEM adoption,” said Gaisford. “Hospitals and physicians are leading the charge here, especially for surgical planning. But medical OEMs are just learning about the ways 3D printed models can enhance device testing and physician education beyond existing modalities of human cadavers and animals, which have significant limitations. With cadavers, the parts have been processed and no longer retain living tissue characteristics and it is often difficult to find a cadaver that has the target pathology within it. Animal models only approximate human anatomy and also lack disease. Many medical OEMs have not stayed up to date with developments in materials and printer capabilities that enable 3D printed models to mimic clinical scenarios more accurately than in the past and to match color realism.”
“Our cleared Materialise Mimics software provides clinicians with the ability to plan and print anatomical models for highly complex cases,” said Crutchfield. “These anatomical models and surgical plans allow for a comprehensive diagnosis and treatment plan that shortens procedure times and improves outcomes. However, adoption has been slowed by lack of reimbursement by benefit providers who have yet to catch up with the technology.”
Hurdles to Larger Adoption
As with any emerging power, 3D printing obviously can’t grow unchecked. Though some believe its abilities could be limitless, medical applications for 3D printing still remain in the adolescent stage. This isn’t altogether unexpected, as some medical applications—bio printing being one example—require 3D printing to boldly go where it isn’t quite ready to go just yet. 3D printers will eventually have to adapt to handle multiple materials, biological components, and different sets of software solutions to accomplish what particularly progressive OEMs hope to achieve.
“Right now, materials capability is the greatest hurdle. Customers are asking for materials that are as hard as bone or soft as skin from the same printer,” said Gaisford. “That’s an incredible technical challenge to achieve. It’s something we are working very hard to improve. The other hurdle is the ease of use of going from concept to printed part. For example, to take an MRI/CT to printed part can take multiple software packages and trained expertise. We need to find ways to better streamline that workflow.”
“The capability for ‘individual’ or ‘custom-bespoke’ manufacturing capabilities requires a more comprehensive data collection and tracking for each patient; a process some manufacturers might not want to take responsibility for,” noted Somers. But he also offered a potential solution to this hurdle, suggesting that “instead, intermediary companies can be used within the supply chain—by either medical groups offering a wide range of services to monitor it for their ‘subscribers,’ or even insurance companies, who mostly pay for them.”
Crutchfield also offered advice on the best way to advance 3D printing’s practicality—and it’s no surprise that the brunt of that work lies with the industry’s friendly neighborhood regulatory body.
“Finalization of the FDA Draft Guidance will provide for a stable basis for companies to build from going forward in implementing additive manufacturing solutions in the market,” he suggested.
Somers expressed a similar view on the most practical way to leap over the hurdles of wider medical 3D printing adoption—aptly navigating any regulatory complexities throughout the entire manufacturing process.
“Since the sea change at FDA from ‘rule-based’ to ‘risk-based’ strategies associated with inspection and evaluation criteria, the onus has been shifted to the manufacturers to provide the statistical data and analyses for their products to be reviewed and monitored,” he explained. “I would apply a similar scenario to the 3D manufacturing: if the products pass the ‘benefit-risk’ test and data is provided ‘as requested’ regarding their functional and operational history, there shouldn’t be any interference with proprietary processes by the FDA.”
Further, Crutchfield explained the importance of payers’ roles in the technology’s wider adoption. “Additionally, acceptance of new 3D printing solutions in the surgical guide and anatomical market as reimbursable items by Medicare/Medicaid and benefit providers is needed.”
Though more a byproduct than a hurdle of larger 3D printing adoption, Nichols stressed the importance of cybersecurity—a topic that’s certainly at the front of the minds of those in the medtech industry who have been paying attention to recent claims of vulnerabilities (whether they are founded or not) in St. Jude Medical Inc.’s implantable cardiac devices.
“Privacy and security will be important considerations as patient-specific data will need to be inputted and transmitted safely,” she advised. “No company is immune to a cyber-extortion scheme, virus, or other malware that could affect their manufacturing systems. In addition, verifying the safety and efficacy of the products that are being manufactured using 3D printing in medtech will be imperative.”
What’s Next on the Queue?
It seems strange to ask where the future of 3D printing is headed when the technology is already so futuristic. But those involved in additive manufacturing, whether providing printing services or developing devices, are always looking ahead to stay on top of the fertile ground 3D printing has laid for medtech development.
“I think in the next five years, we will see new materials with greater clearances and certifications for use in production parts,” envisioned Gaisford.
“The medical industry will continue to push the boundaries of additive manufacturing in the future, as the industry drives towards cost-effective, patient-specific solutions that address each unique condition,” predicted Crutchfield. “As we work with our workflow solution partners we see several areas developing in the next five years, such as patient-specific metal implants, resorbable implants, bio printing, and 3D printed pharmacology—just to name a few.”
“The industry is seeing an increase in the number of ‘point of care’ device manufacturing, where 3D printers are now located in the hospital where a patient’s specific part can be manufactured on the spot,” Nichols noted. But she also brought up an important point regarding who is truly “responsible” for a device that is manufactured in this way. “This raises an interesting question from a products liability perspective, as the lines are blurred on who truly is the ‘manufacturer’ of the final medical product—is it the 3D manufacturer, the hospital, or the physician?”
Somers was optimistic in his forecast, saying, “3D printing will continue to move ever upward.” He was also hopeful for additional next-generation technologies that could one day companion products manufactured by 3D printing to develop treatments that healthcare professionals might never have dreamed of. “[3D printing], along with nanotechnology and other biological process therapies, are revolutionizing the medical treatment field, all for the benefit of patients.”