Maria Shepherd, Medi-Vantage09.08.16
The four types of atrial fibrillation (AFib) that are treatable by ablation are persistent, longstanding persistent, paroxysmal, and permanent AFib.1 Current treatments for AFib are cardioversion, prescription medication for management of rate and/or rhythm control, and, when all else fails, surgery or catheter ablation. A single ablation procedure may not consistently eliminate AFib and repeat ablations are sometimes required to eradicate this disease state.
Why This Is Important
AFib is one of the leading causes of stroke and it is estimated that patients with AFib have a five-fold increased risk of stroke.2 This makes the treatment of AFib a significant challenge to global healthcare systems. It is estimated that greater than 750,000 U.S. hospital admissions occur each year because of AFib, and that AFib is the cause of an estimated 130,000 deaths each year.3,4 The annual cost to the United States for AFib is approximately $6 billion. Patients with AFib incur approximately $8,705 in additional medical costs per year than patients without AFib.5,6
Catheter Ablation
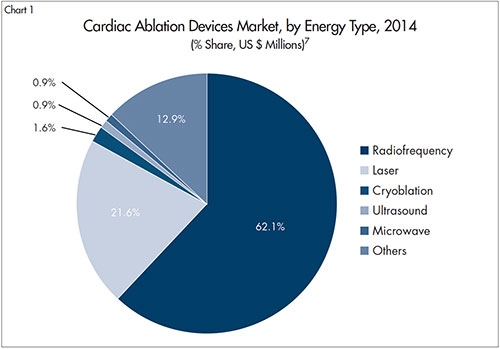
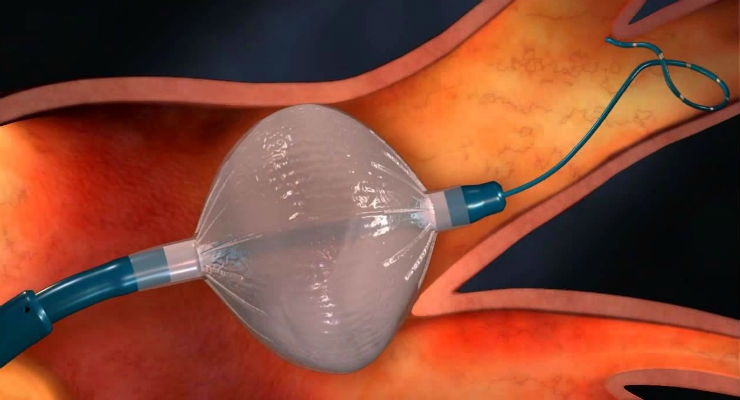
Catheter-based ablation procedures are mainly segmented by three types of technologies: radio-frequency ablation, balloon ablation, and cryoablation. Radio-frequency ablation is currently the market leader, with an estimated 60 percent share of the market7 (Chart 1).
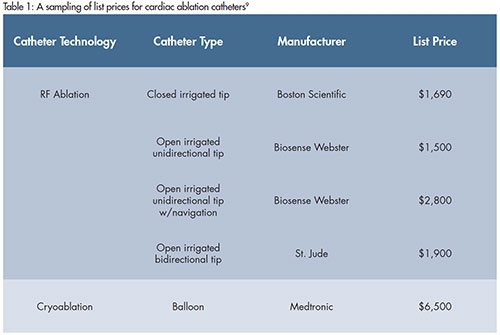
For good reason, there have been many acquisitions in the cardiac catheter ablation space. The global addressable market opportunity is large and estimated to reach $1.7 billion by 2018.8 This market is growing rapidly and these catheters (and their required accessories) command premium prices (Table 1).
Conclusion and Recommendations
Hospitals continuously assess medical technology to improve clinical outcomes and sustain their own financial health. Despite the high prices and growing market share, cardiac ablation procedures are not ideal. Electrophysiologists report that average procedure times are greater than two hours, which means that fluoro exposure is also high. Depending upon which study referenced, cardiac ablation success rates—where the patient remains AFib free at one year—still only average 60 percent.
Companies serving the medical specialty of cardiology need to explore product design changes in the race to market to address these challenges. These design innovations will strengthen device companies’ medtech economic value proposition.
References
Maria Shepherd has more than 20 years of leadership experience in medical device/life-science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical where she boosted the company valuation prior to its acquisition by Covidien/Medtronic, director of marketing for Philips Medical, and senior management roles at Boston Scientific Inc., she founded Medi-Vantage. Medi-Vantage provides marketing and business strategy and innovation research for the medical device industry. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services and assesses prospective acquisitions. Shepherd has taught marketing and product development courses and is a member of the Aligo Medtech Investment Committee (www.msbiv.com). She can be reached at 855-343-3100 x 102 or at mshepherd@medi-vantage. Visit her company’s website at www.medi-vantage.com.
Why This Is Important
AFib is one of the leading causes of stroke and it is estimated that patients with AFib have a five-fold increased risk of stroke.2 This makes the treatment of AFib a significant challenge to global healthcare systems. It is estimated that greater than 750,000 U.S. hospital admissions occur each year because of AFib, and that AFib is the cause of an estimated 130,000 deaths each year.3,4 The annual cost to the United States for AFib is approximately $6 billion. Patients with AFib incur approximately $8,705 in additional medical costs per year than patients without AFib.5,6
Catheter Ablation
Catheter-based ablation procedures are mainly segmented by three types of technologies: radio-frequency ablation, balloon ablation, and cryoablation. Radio-frequency ablation is currently the market leader, with an estimated 60 percent share of the market7 (Chart 1).
For good reason, there have been many acquisitions in the cardiac catheter ablation space. The global addressable market opportunity is large and estimated to reach $1.7 billion by 2018.8 This market is growing rapidly and these catheters (and their required accessories) command premium prices (Table 1).
Conclusion and Recommendations
Hospitals continuously assess medical technology to improve clinical outcomes and sustain their own financial health. Despite the high prices and growing market share, cardiac ablation procedures are not ideal. Electrophysiologists report that average procedure times are greater than two hours, which means that fluoro exposure is also high. Depending upon which study referenced, cardiac ablation success rates—where the patient remains AFib free at one year—still only average 60 percent.
Companies serving the medical specialty of cardiology need to explore product design changes in the race to market to address these challenges. These design innovations will strengthen device companies’ medtech economic value proposition.
References
- http://bit.ly/data091601
- http://bit.ly/data091602
- Agency for Healthcare Research and Quality. Weighted national estimates. HCUP National Inpatient Sample [online]. 2012. http://bit.ly/data091603. Accessed August 12, 2016
- Centers for Disease Control and Prevention. About multiple cause of death 1999–2011. CDC WONDER Online Database. 2016. http://bit.ly/data091604. Accessed August 12, 2016
- January CT, et al. 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation. JACC. 2014;64(21):2246–80. 2.
- Mozaffarian D, et al. Heart disease and stroke statistics—2015 update: A report from the AHA. Circulation. 2015;131:e29–e322.
- http://bit.ly/data091605
- BCC, Ablation Devices: Technologies and Global Markets, 2014
- J Interv Card Electrophysiol. 2013 Mar; 36(2): 157–165.
Maria Shepherd has more than 20 years of leadership experience in medical device/life-science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical where she boosted the company valuation prior to its acquisition by Covidien/Medtronic, director of marketing for Philips Medical, and senior management roles at Boston Scientific Inc., she founded Medi-Vantage. Medi-Vantage provides marketing and business strategy and innovation research for the medical device industry. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services and assesses prospective acquisitions. Shepherd has taught marketing and product development courses and is a member of the Aligo Medtech Investment Committee (www.msbiv.com). She can be reached at 855-343-3100 x 102 or at mshepherd@medi-vantage. Visit her company’s website at www.medi-vantage.com.